View sample leprosy research paper. Browse research paper examples for more inspiration. If you need a health research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Introduction
Leprosy, although rarely fatal, occupies a unique place in the field of human disease. The earliest descriptions go back thousands of years. Leprosy was the first infectious disease for which the causative organism was discovered; yet it still eludes cultivation on artificial media (Rees and Young, 1994). The stigma attached to leprosy has caused untold suffering for countless numbers of people, even the word ‘leprosy’ has become a curse word in some languages. Today, stigma continues to hinder early presentation of new cases and adherence to treatment, and to threaten the social integrity of the people affected in most countries where leprosy is endemic. Leprosy is both a dermatological condition and a chronic disease of the peripheral nervous system. Management of leprosy and its complications involves village health workers, paramedical workers, dermatologists, neurologists, physicians, surgeons, public health experts, physiotherapists, occupational therapists, nurses, social workers, and, above all, the people affected by leprosy themselves.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
Epidemiology
Etiology
Leprosy is an infectious disease caused by Mycobacterium leprae (M. leprae). M. leprae was the first human microbial pathogen to be discovered, by Dr. G. H. Armauer Hansen in Norway in 1873 (Rees and Young, 1994). A Gram-positive, strongly acid-fast, rod-shaped bacteria, M. leprae measures 1–8 mm by 0.3 mm. Despite being the first pathogen to be identified, it still cannot be cultured on artificial media. However, M. leprae will multiply when inoculated in mouse footpads, immune-compromised mice, or nine banded armadillos. The generation time of 11 to 13 days is uniquely slow even among the slow-growing mycobacteria (the generation time of M. tuberculosis is 20 hours). M. leprae is the only bacterium that infects peripheral nerves. It is an obligate intracellular parasite, predominantly residing in macrophages and Schwann cells, particularly of unmyelinated nerve fibers (Rees and Young, 1994).
Case Definition
Because of the chronicity of leprosy, it is often impossible to determine the exact onset of disease. Effective antileprosy treatment usually causes the typical skin and nerve lesions to regress, but often patients still have obvious signs and symptoms of leprosy at the end of treatment, such as residual hypopigmentation of former skin lesions, sensory or motor impairment, or secondary impairments. However, the World Health Organization (WHO) decided that a patient who is cured from a bacteriological point of view should no longer be called a ‘case.’ A case was thus defined as ‘‘any person clinically diagnosed as having leprosy, with or without bacteriological confirmation of the diagnosis, and needing chemotherapy.’’ Patients who have completed chemotherapy are therefore removed from the register and are no longer counted in the official statistics. However, many have residual impairments needing continued attention to prevent further deterioration, and a proportion will continue to develop new impairments and disabilities even after successfully completing chemotherapy (Croft et al., 2000). Thus, although no longer ‘cases,’ these people still need appropriate care, including services to help prevent disability. In addition, there are people who suffer the consequences of leprosy in terms of activity limitations or restrictions in (social) participation (van Brakel, 2000). They are no longer ‘leprosy cases,’ but may still be in need of rehabilitation services, for example, community-based rehabilitation or vocational training. These groups of people need to be taken into account during health services planning exercises.
Occurrence
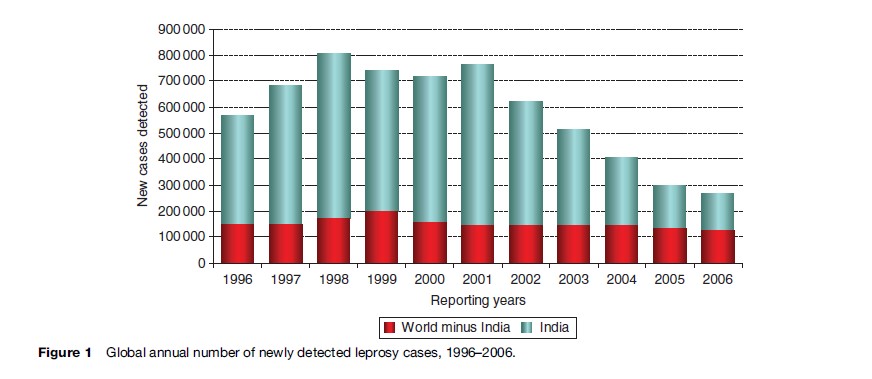
WHO regularly reports data on registered prevalence and new case detection from the various WHO regions and countries. It is known that these data are affected by several operational factors, such as changes in case definitions, the practices for case detection, treatment, and registration. Global reported annual detection reached a peak of 804 357 in 1998, before falling to around 621 000 in 2002 and 515 000 in 2003. This downward trend continued during 2004 and 2005, with 408 000 and 299 000 newly detected cases, respectively. The sharp decline in the number of annually reported new cases over the last four years can be almost fully attributed to the reduction in case detection in India alone (Figure 1). It is unclear at the time of writing whether this reflects a true decline or whether it is mainly a reflection of operational factors. The reported proportion of ‘grade 2 disability’ among new leprosy patients in different parts of the world ranges between 3% and 32%. In 1997, WHO estimated the global prevalence of (ex-)patients with visible impairments (WHO disability grade 2) to be 2 million. This estimate would be even higher if people who only have sensory impairment were included. In some countries and areas the prevalence of people with residual impairments is high. This places a considerable demand on the health services regardless of the new case detection rate.
Reservoir
The primary reservoir of infection is human beings. Naturally acquired infection has been reported in nine-banded armadillos, chimpanzees, and mangabey monkeys, but these are not currently considered a source of infection of any significance. However, the fact that the incidence so far failed to decrease significantly in many countries has led to speculation regarding other possible reservoirs of M. leprae, such as soil and water. New techniques, such as polymerase chain reaction (PCR), have made more sophisticated investigations of environmental sources possible and new studies are under way.
Mode Of Transmission
The main mode of transmission is believed to be droplet infection by bacilli excreted from the nasal cavity of untreated lepromatous (or other multibacillary) patients. Skin to skin transmission remains a possibility, although it is doubtful that bacilli could enter intact skin. The most likely portal of entry is believed to be the upper respiratory tract.
Incubation Period And Communicability
The incubation period is usually between 2 and 5 years but ranges from a few months to more than 20 years. It is not known at what stage an infectious patient starts excreting bacilli, nor how long this would continue in the absence of treatment. Effective antileprosy treatment, for example with rifampicin, usually renders a patient noninfectious within a few days.
Susceptibility
The factors that determine the susceptibility of individuals to leprosy are still not known. Prolonged and close contact with a known patient, particularly if this patient is of the lepromatous type, significantly increases the risk of becoming a case (Fine et al., 1997). However, it has been shown that others – household contacts, neighbors, and contacts at work – are also at increased risk of developing leprosy (Bakker et al., 2006; Moet et al., 2006). Susceptibility to the disease may be increased by prolonged exposure to M. leprae, but this remains hypothetical and difficult to prove. The type of leprosy to develop in susceptible individuals may be determined, at least in part, by human lymphocyte antigen (HLA)-linked genes. There is evidence that leprosy is highly infectious, but that only a small proportion of infected individuals develop clinically overt disease. Recently, two genes were identified that have a strong association with increased susceptibility. Further studies are under way.
Diagnosis, Classification, And Treatment
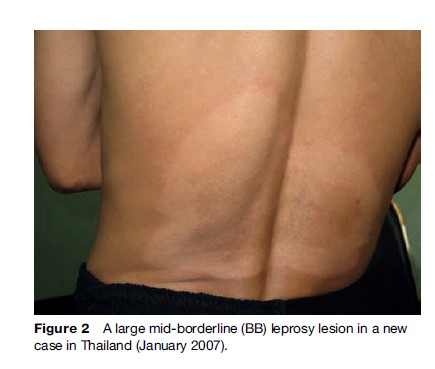
The disease spectrum of leprosy varies from a single self-healing hypopigmented macule to a generalized illness that causes widespread peripheral nerve damage and affects even bones and internal organs. Skin lesions may be either well or ill-defined hypopigmented macules, plaques, or nodules, localized in a particular area or symmetrically distributed over the whole skin (Figure 2). They may be hypesthetic, anesthetic, hyperesthetic (paresthesia), or have normal sensibility. Nerve lesions occur in dermal nerves as well as in superficial sensory nerves and mixed nerve trunks. One or more nerves may be enlarged on palpation. Commonly affected are the greater auricular, radial (cutaneous), ulnar, median, common peroneal, sural, and posterior tibial nerves. Other nerves that may be affected are the facial nerve, giving a characteristic paresis or paralysis with lagophthalmos, usually without affecting the oral branch, and the corneal branches of the trigeminal nerve, causing corneal hypesthesia or anesthesia. Secondary signs such as clawing of fingers and toes, ‘absorption’ of digits due to repeated injury, and dry skin are due to impairment of motor, sensory, and autonomic nerve function. In patients with long-standing ‘lepromatous leprosy,’ madarosis, and nasal collapse occur, but are now uncommon.
Diagnosis
The diagnosis ‘leprosy’ is based on finding at least one of three so-called cardinal signs: diminished sensibility (detected with cotton wool and/or a nylon filament) in a typical macule or plaque in the skin (Figure 3), palpable enlargement of one or more peripheral nerve trunks at specific sites, and the demonstration of acid-fast mycobacteria in a slit skin smear.
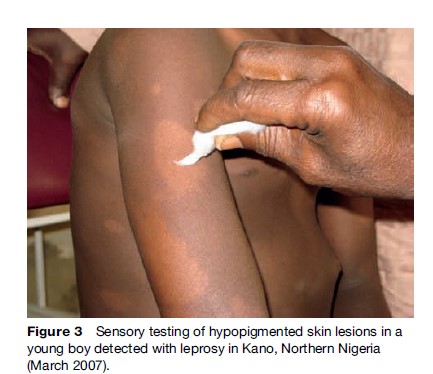
If diminished sensation cannot be clearly demonstrated, the diagnosis cannot be made based on the skin lesion(s) alone. Palpable thickening of peripheral nerve trunks due to causes other than leprosy is rare. The differential diagnosis of nerve thickening includes hereditary diseases such as Charcot Marie Tooth disease and De´jerine Sottas disease, amyloid neuropathy, and post-traumatic neuritis. These are generally considered to be so rare that in a leprosy-endemic area the finding of ‘definite’ enlargement of a peripheral nerve trunk is sufficient to establish the diagnosis of leprosy. However, one should be careful before diagnosing leprosy on the basis of a single enlarged nerve, particularly in the case of ‘pureneuritic’ leprosy in which there are no characteristic skin lesions and the skin smear is negative.
Diagnosis may be confirmed by histology of affected skin or nerve tissue, although the histological diagnosis of biopsies from early suspected lesions is not straightforward. For the bacteriological examination of a (suspected) leprosy patient, usually four to six slit skin smears are taken from different sites on the body. These should include both earlobes and, where possible, at least two active-looking skin lesions. The smears are fixed and then stained with the Ziehl-Neelsen method. The results are reported using the bacteriological index (BI), a 7-point scale based on the number of bacilli present per oil immersion field; either the highest or the average score of the individual smears is used (Rees and Young, 1994). It should be noted that only a relatively small proportion of all cases have positive smears, so this test can never be used to rule out leprosy.
Immunology And Classification
The various clinical manifestations are the result of the immune response of the host to infection with M. leprae. In the 1960s, Ridley and Jopling proposed a 5-group classification system based on immunological and histological characteristics. This system is still used in research. Patients with limited disease (one or a few skin lesions only) and a potent cell-mediated immune (CMI) response against M. leprae were called ‘tuberculoid’ (TT), while those with extensive disease and an absent CMI response were called ‘lepromatous’ (LL). Between these two ends of the immunological spectrum came the socalled borderline patients (BT, BB, or BL). In 1982, WHO introduced a 2-group classification system based on the probable number of M. leprae present. Patients assumed to harbor only a few bacilli are called ‘paucibacillary’ (PB), while those assumed to harbor many are called ‘multibacillary’ (MB). From 1998 on, classification would be done on clinical signs only, but skin smear results are taken into account when available. Patients with more than five skin lesions or who are BI-positive are classified as ‘MB,’ while all others are classified as ‘PB.’
Treatment
Sulphone therapy for leprosy was introduced in the late 1940s. This was successfully used as monotherapy for two decades. However, in the 1970s, resistance to dapsone emerged on a wide scale. To counter this serious problem, WHO introduced MDT in 1982. PB patients were to be given a 6-month regimen (PB MDT) consisting of daily dapsone (DDS, 100 mg once daily) and monthly rifampicin (600 mg once a month, supervised). MB patients were to be treated with a three-drug MDT regimen for a minimum of 2 years, but where possible until the skin smear had become negative. Besides the drugs of the PB regimen, the MB regimen includes clofazimine 50 mg daily and 300 mg once monthly, supervised. The results of these regimens have been very good with very few relapses. The duration of MB MDT was shortened in 1993 to a fixed-duration 24-month regimen and, in 1998, to a 12-month regimen. MDT has been very successful in the treatment of individual cases, but the epidemiological impact has been less striking; registered prevalence has declined because of the reduced duration of treatment, but disease transmission does not seem to have been greatly reduced, presumably because significant transmission occurs in the late stages of incubation, prior to diagnosis and the start of treatment.
Complications
The skin signs of leprosy are relatively harmless, but complications of the disease may lead to severe consequences, such as blindness, infertility, and severe sensory and motor disability. Most of the complications are the result of immune-mediated damage to the peripheral nervous system. This may be insidious, as part of the ‘normal’ pathophysiological process of leprosy, in which granulomatous inflammation destroys subcutaneous nerves. But much more dramatic and debilitating are the so-called leprosy reactions. Other complications include destruction of the soft palate, causing palatal perforation, and bone changes due to infiltration with M. leprae. These occur only in advanced cases and are most common in sites where the temperature is below 37 degrees, such as the nasal cavity and the fingers and toes. Destruction of the nasal septum leads to the typical collapsed or ‘saddle’ nose. Dermatological complications include madarosis and skin wrinkling, occurring only in lepromatous patients. Most ocular complications are secondary to iritis or lagophthalmos. Corneal anesthesia may be present following infiltration of M. leprae into the corneal branches of the trigeminal nerve. This may lead to eye injury due to failure of the blink reflex.
Reactions And Neuritis
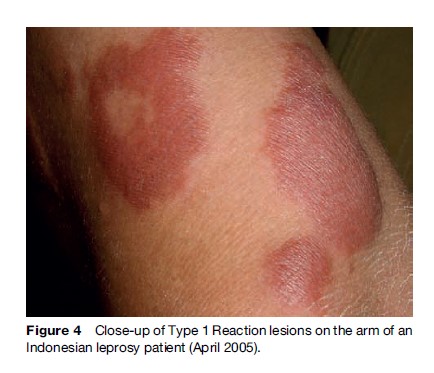
The term ‘reaction’ is used to describe the appearance of signs and symptoms of acute inflammation due to hypersensitivity to leprosy antigens. Clinically there is redness, swelling, heat, and sometimes tenderness of skin lesions, and there may be swelling, pain, and tenderness of nerves, often accompanied by loss of function. New skin lesions may appear. The nerve function impairment is likely to become permanent if left untreated. Two types of reaction are distinguished. A Type I Reaction (T1R) or ‘reversal reaction’ may occur in both PB and MB patients. A T1R is a delayed-type hypersensitivity (DTH) reaction involving the cell-mediated immune system (Britton and Lockwood, 1997). The localized DTH response causes local inflammation of skin and nerves at the site where antigen is present. Erythema nodosum leprosum (ENL) or Type II Reactions occur in MB patients only (especially those with ‘lepromatous’ leprosy). ENL is a systemic immune complex reaction, in which antibody–antigen complexes may be deposited in various tissues, leading to vasculitis (Britton and Lockwood, 1997). At the time of diagnosis, during drug collection visits, and at release from treatment (RFT), all patients must be questioned and examined for signs and symptoms of reactions (Figure 4).
Not infrequently patients develop nerve damage without any of the obvious signs of the two types of reactions outlined earlier. This is called ‘silent neuritis’ or ‘silent neuropathy’ and is defined as ‘‘any sensory and/or motor impairment of recent onset (<6 months duration) without spontaneous symptoms of nerve pain or tenderness or signs of a reaction (T1R or T2R).’’
Peripheral Neuropathy
M. leprae is the only bacillus known to selectively invade human peripheral nervous tissue. A histological picture of acid-fast bacilli inside the peripheral nerve is considered pathognomonic for leprosy. From the point of view of the pathology of leprosy, Ridley and Job wrote ‘‘Leprosy is essentially a disease of the peripheral nerves.’’ The question is therefore not whether or not the patient has neuropathy, but whether the neuropathy is severe enough to be detected with the instruments available. However, only clinically detectable neuropathy requires treatment. There is evidence that mild sensory neuropathy often heals without treatment other than MDT.
Damage of sensory, motor, and autonomic nerve fibers is common. Motor nerve impairment has long been recognized as an important problem in leprosy, because it often leads to visible impairment (deformity). Regular testing of voluntary muscle strength was already suggested in the 1960s. It has been adopted on a worldwide scale as a measure of neural function and is often the only outcome parameter reported. Although the importance of motor function in daily life is beyond dispute, the importance of sensory function is often underestimated. A patient with insensitive feet is at constant risk of injury, while those with insensitive hands often have considerable activity limitations (van Brakel and Anderson, 1997). Loss of protective sensation in eyes, hands, and feet is responsible for much of the long-term morbidity caused by leprosy. Autonomic nerve damage is important because the resulting dryness of the skin, and possibly also the changed microvascular physiology, are additional risk factors for injury.
Treatment Of Reactions And Nerve Function Impairment
Patients who only have skin signs of reaction, without ulceration of the lesions and without signs of nerve involvement, may be treated with rest and anti-inflammatory drugs such as aspirin (600 mg three times daily for 14 days) or chloroquine phosphate (250 mg three times daily for 14 days). Patients with a mild reaction should be reviewed carefully by a doctor or supervisor after 2 weeks for possible worsening of the reaction, particularly the occurrence of nerve function impairment (NFI). If the reaction is still mild, the above treatment should be continued for another 2 weeks, after which the patient is reviewed again. Chloroquine should not be given for more than 4 weeks.
If there are signs of a severe reaction (the presence of NFI, ulceration, severe edema, or the involvement of other organs such as eyes or testes), or if a mild reaction fails to improve within 4 weeks, prednisolone should be prescribed. The patients with certain (relative) contraindications must first be referred to a specialist center. These include intercurrent infections, suspected nerve abscess, painful red eye, any hand or foot wound, suspected peptic ulcer, cough for more than 3 weeks (suspected tuberculosis), suspicion of other conditions such as hypertension, diabetes, and pregnancy, or age of 15 years or less.
Steroid Regimens
Further studies to determine the optimal dose and duration of the steroid treatment of reactions are still needed. We currently recommend the following prednisolone regimen for patients of 15 years or older:
- Starting dose 40 mg, single dose per day, given in the morning. This corresponds to about 0.5–1 mg per kilogram body weight for most patients.
- Tapering 5 mg every 2 weeks for PB patients and every 4 weeks for MB patients. The total duration of a standard course would be 12 weeks for PB and 24 weeks for MB.
- If the dose needs to be increased for any reason during the steroid course, this should be done in steps of 10 mg per week. Reasons for this include failure to control the acute signs and symptoms of the initial reaction, including sensory and/or motor impairment of recent onset, recurrence of acute signs and symptoms, and increasing sensory or motor deficit.
- Sometimes the patient has to be admitted during the first 2 to 4 weeks or until the signs and symptoms of reaction have begun to subside.
Regimen for children under 15 years of age:
- Starting dose 30 mg, single dose per day, given in the morning for the first week, then 30 mg on alternate days for 1 week; then
- Reduce the dose to 25 mg on alternate days, tapering 5 mg every 2 weeks; total duration of a standard course: 12 weeks.
The rationale behind this regimen is that suppression of the pituitary and adrenal glands is unlikely to happen within 1 week, while the initial beneficial effect of the steroids can be expected within the first 1 to 2 weeks.
Nerve Function Assessment
Autonomic impairment is common in leprosy, but is difficult to test. Assessing dryness with the fingers or back of the hand is a simple but crude test, which can be difficult to interpret on the foot soles of people walking barefoot or wearing open sandals, particularly in the dry season.
An appropriate test of touch sensation is skin indentation. The most widely used sensory test in the field of leprosy is the ballpoint pen test. Its strengths lie in its simplicity and in the almost universal availability of the instrument. The test is not sensitive enough to pick up mild impairment, but is a valid test of protective sensation, provided the correct technique is used. The principle of sensibility testing with graded filaments, originally introduced by von Frey in the late nineteenth century, was revived by Semmes and Weinstein using standardized nylon monofilaments. The test method and ways to record and interpret results are described elsewhere (Figure 5).
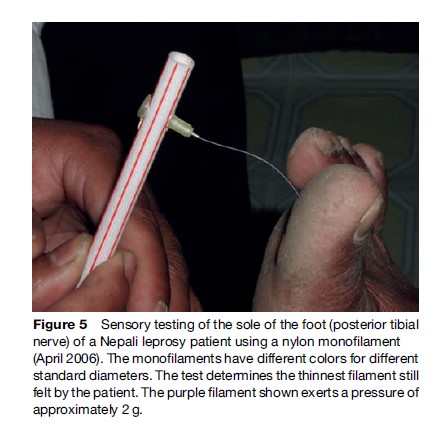
The voluntary muscle test (VMT) has been used widely in leprosy since 1968. In the early 1980s, a 6point scale was recommended for grading the test when assessing people affected by leprosy. Shorter 3 and 4-point scales are also in use. The most commonly used is the 3-point scale which grades muscle strength as ‘strong,’ ‘weak,’ or ‘paralyzed.’ The VMT is not easy to perform technically, as the tester has to use ‘intrinsic normal values’ based on his or her experience of normal muscle strength. Normal muscle strength varies with sex and age, so this requires considerable practice. An advantage of the VMT is that no testing instruments are required. Nerve function assessment should be done at the first clinic visit of each patient and at least every 3 months during MDT treatment.
Long-Term Consequences Of Leprosy
When detected and treated in time with corticosteroids, primary impairments may be reversible. However, a substantial proportion of patients (10–50%) do not recover or get worse. Among new patients, 3% to 32% present with secondary impairments (grade 2 disability), such as wounds, contractures, and shortening of digits, usually preventable consequences of the autonomic, sensory, and/or motor neuropathy. All people with impairments need careful and repeated teaching on methods to prevent further impairment and subsequent disability. Following such impairments many people experience limitation of activities (of daily living). As a result of such limitations, or because of the stigma attached to visible impairments or even to the diagnosis ‘leprosy,’ many people have socioeconomic problems. They are restricted in their (social) participation (van Brakel, 2000). A large, but unknown percentage of people succeed in overcoming these activity limitations and participation restrictions by themselves and do not need outside help, in spite of residual impairments. A certain percentage, however, do need rehabilitation assistance, including physical or occupational therapy, reconstructive surgery, or temporary socioeconomic assistance.
Prevention Of Disability
Prevention of disability (POD) is a key objective in any leprosy control program. The main components are:
- Primary prevention. Early detection of leprosy and adequate treatment are by far the most effective measures to prevent disability (Meima et al., 1999). However, since 10% to 20% of patients will develop new sensory or motor impairments during or even after MDT, early detection of reactions and NFI and subsequent steroid treatment are also of vital importance. The Consensus Conference on Prevention of Disability in Cebu, Philippines (2006), highlighted the current challenge to make this treatment widely available, as close to where patients live as possible; if the treatment is to be made available, it is essential that patients themselves are aware of the possible symptoms that may occur, and that local health workers have the basic knowledge and skills to assess the problem and then either treat or refer (WHO, 2006).
- Secondary prevention. If people have already developed irreversible neural impairment or even secondary impairments (such as wounds and contractures), one needs to prevent these from getting worse. Reconstructive surgery plays a part in secondary prevention, since the goal is often to prevent further injury. Protective footwear for people with insensitive feet is sometimes provided by the program, but often people must buy the footwear themselves. Other ‘assistive devices’ may correct or prevent activity limitations.
The main strategy for secondary prevention, however, is ‘self-care’: teaching the affected persons how to look after their damaged eyes or limbs, how to prevent injury, and how to treat wounds. They will have to do this conscientiously for the rest of their lives. Self-care groups can be an effective means to support this. It is now realized that effective self-care depends largely on the empowerment of those affected, so that they are motivated and enabled to pursue their own POD goals (WHO, 2006) – in much the same way as is happening in the management of other chronic diseases.
Tertiary prevention. The objective is to prevent participation restrictions, often resulting from the stigma attached to leprosy. Strategies for prevention therefore include counseling of the affected person and their families, neighbors and community, vocational training, and advocacy work (Gershon and Srinivasan, 1992).
Rehabilitation
People suffering from activity limitations or participation restrictions may need rehabilitation services. If at all possible, these should be sought within existing facilities for people with disability, such as community-based rehabilitation programs, counseling services, or vocational training schemes. Reconstructive surgery has an important role in improving the function of hands and feet and in reducing stigmatizing deformities. Promoting the dignity of people affected by leprosy is one of the most important tasks in health education and advocacy work. Leprosy ‘treatment’ is only successful if, in the end, people can function optimally and are accepted as equal members of their society. Organization of people affected by leprosy in self-help groups or organizations can be a powerful way to promote rehabilitation and to fight against stigma and discrimination.
Leprosy Control
Disease control can be defined as the reduction of the incidence and prevalence of the disease, and of the morbidity and mortality resulting from the disease to a locally acceptable level as a result of deliberate efforts. Continued intervention is required to maintain the reduction.
The objectives of leprosy control are:
- To interrupt transmission of the infection, thereby reducing the incidence of disease
- To treat patients in order to achieve their cure
- To prevent the development of associated deformities
- To achieve complete rehabilitation.
The strategy to achieve control of leprosy consists of three major elements:
- Early case detection
- Adequate chemotherapy (MDT)
- Provision of high-quality, comprehensive care for the prevention of disabilities and rehabilitation of patients.
Comprehensive care includes early diagnosis and treatment of reactions, eye care, wound and ulcer treatment, patient education, self-care, use of protective footwear and other protective devices, physiotherapy, reconstructive surgery, and socioeconomic rehabilitation (ILA, 2002; WHO, 2006).
Implementation of this strategy ideally requires readily accessible, efficient, and sustainable health services that cover the population fully, and that are accepted by the community and the patients. This strategy implies that leprosy control activities should be implemented by the general health services (ILA, 2002).
Prevention
The objective of communicable disease control is ultimately to stop transmission of the causative agent, so that no new individuals will get infected and be at risk of developing disease. Disease may be prevented by preventing transmission or by preventing an infected person from developing the disease (e.g., through vaccination). In some diseases, such as leprosy, HIV, or typhoid, it may be very difficult to prevent transmission, because people may be asymptomatic carriers who are themselves unaware that they are infected and infectious to others. No methods are available to prevent infection with M. leprae. Isolation of infectious patients is no longer practiced, for two main reasons: Most patients have been infectious for a long time by the time they are detected and therefore transmission has likely already taken place; and treatment with MDT renders even a lepromatous patient noninfectious within a few days. MDT can therefore be seen as ‘chemical isolation.’
Prevention of disease in infected persons is also difficult because as yet no reliable tests exist to detect (subclinical) infection. Some authorities recommend rifampicin prophylaxis for use in close contacts of lepromatous patients. Two large field trials in Indonesia and Bangladesh have shown that one or two doses of prophylactic rifampicin reduces the risk of leprosy among contacts by 50% after 2 years (Bakker et al., 2006; Moet et al., 2006). There is now substantial evidence that (repeat) vaccination with BCG gives up to 75% protection against the occurrence of clinical leprosy (Fine and Smith, 1996). However, no policy for revaccination of at-risk people has yet been recommended. A case could be made for BCG (re)vaccination of contacts of MB leprosy cases, at least where the prevalence of HIV is still low.
The only means to reduce transmission of leprosy is to render infectious cases noninfectious as early as possible. Therefore, early case finding and treatment of all cases with MDT are the two main methods for the control of leprosy as a communicable disease.
Elimination Of Leprosy
In 1991, the World Health Assembly declared the ‘elimination of leprosy as a public health problem’ as a goal to be achieved by the year 2000. ‘Elimination’ was defined as a prevalence of less than one case per 10 000 population. Underlying the elimination strategy was the hypothesis that, because leprosy patients are assumed to be the sole source of infection, early detection and treatment with MDT would reduce transmission of M. leprae. Once the prevalence fell below this level, the chain of transmission was expected to be broken, and leprosy would disappear naturally. However, despite a dramatic decline in registered prevalence rates over the past two decades, case detection rates (CDR) have remained more or less stable or have declined at a much slower rate over the past 10 to 15 years in most endemic countries (Figure 1) (ILA, 2002). Prevalence is dependent on duration of disease and incidence. Over the past decade, the duration of treatment – and thus, effectively, the duration of disease – has been shortened, leading to dramatic but artificial reductions in prevalence. Several authors have therefore argued that prevalence and prevalence rates are no longer adequate indicators to monitor the epidemiological situation of leprosy in a given country or area (ILA, 2002). CDRs should be interpreted in conjunction with other indicators, such as the child proportion and the proportion of cases with grade 2 disability among new cases (ILA, 2002). Recently, WHO has also identified these indicators as important tools for monitoring and evaluation of the Global Strategy 2006–2010 (WHO, 2006).
The elimination target and the tremendous effort of the WHO in trying to achieve this target have had a major impact on leprosy control worldwide. Political commitment in endemic countries has increased greatly. MDT services have expanded to cover the majority of the population and almost 100% of new cases are now treated with MDT. As a result, the prevalence of cases on treatment has been reduced by over 90%. On the negative side, the elimination target has led many to believe that leprosy would no longer be a problem after AD 2000, leading to a decrease in funding and research efforts. Others believed elimination to be the same as eradication and, as a consequence, believed leprosy would soon be history. Governments were led to believe that leprosy services would no longer be needed after elimination had been achieved, jeopardizing the sustainability of leprosy control programs. In the meantime, the elimination target has been reached on a global scale, but in several major endemic countries, the target was postponed until 2005.
The current high proportion of children among new cases (13%) indicates continued transmission. This, in combination with the long incubation period of leprosy, means that new cases will continue to appear for many years to come (ILA, 2002). Effective leprosy services, capable of providing adequate standards of care under low-endemic circumstances, need to be sustained (WHO, 2005). There is now general agreement on this, reflected in current plans and strategies, which have been endorsed by all major stakeholders with an interest in leprosy control (WHO, 2005).
Integration Of Leprosy
A key strategy to provide sustainable leprosy control services with a wide geographical coverage is the integration of such services into the general health-care system of a country. Integration has now been implemented or is in progress in most endemic countries. It is important to realize, however, that in situations in which CDRs are low, a focused approach is appropriate, whereby services are provided mainly in selected general health facilities in areas where leprosy still occurs. The skills of peripheral health workers will be limited mainly to suspecting leprosy. Referral centers should verify the diagnosis and start the treatment of the patient. Continuation of treatment could be delegated to the peripheral health facility serving the community in which the patient resides. The community should be informed, and the general health staff of the peripheral health facility should be trained in diagnostic skills and case management (ILA, 2002).
Conclusion
Leprosy is an age-old disease, mainly manifesting itself in the skin and the nerves. It is notorious for the deformities and disabilities it causes. The strong stigma attached to leprosy has caused untold suffering and continues to this day, although there are signs of increasing acceptance of people affected, especially in the general health services. MDT provides an effective cure and is now globally available. New impairments may occur before, during, and after treatment, highlighting the importance of prevention of disability during every stage of the disease. People affected by the long-term consequences of leprosy should be offered rehabilitation services where possible through existing community-based rehabilitation programs. Epidemiological analysis indicates that new cases will continue to appear for many years to come. Therefore, effective, sustainable leprosy control services need to be continued as an integral part of general health services.
Bibliography:
- Bakker MI, Hatta M, Kwenang A, et al. (2006) Risk factors for developing leprosy: A population-based cohort study in Indonesia. Leprosy Review 77(1): 48–61.
- Britton WJ and Lockwood DNJ (1997) Leprosy reactions: Current and future approaches to management. Bailliere’s Clinical Infectious Diseases 4: 1–23.
- Consensus Conference on Prevention of Disability (2006) Consensus statement on prevention of disability. Leprosy Review 77(4).
- Croft RP, Nicholls PG, Richardus JH, and Smith WC (2000) Incidence rates of acute nerve function impairment in leprosy: A prospective cohort analysis after 24 months. The Bangladesh Acute Nerve Damage Study. Leprosy Review 71(1): 18–33.
- Fine PEM and Smith PG (1996) Vaccination against leprosy: The view from 1996. Leprosy Review 67: 249–252.
- Fine PE, Sterne JA, Ponnighaus JM, et al. (1997) Household and dwelling contact as risk factors for leprosy in northern Malawi. American Journal of Epidemiology 146(1): 91–102.
- Gershon W and Srinivasan GR (1992) Community-based rehabilitation: An evaluation study. Leprosy Review 63(1): 51–59.
- International Leprosy Association (ILA) (2002) Report of the International Leprosy Association Technical Forum, Paris, France, 22–28 February. International Journal of Leprosy and Other Mycobacterial Diseases 70(supplement 1): S3–S62.
- Meima A, Saunderson PR, Gebre S, Desta K, van Oortmarssen GJ, and Habbema JD (1999) Factors associated with impairments in new leprosy patients: The AMFES Cohort. Leprosy Review 70(2): 189–203.
- Moet FJ, Pahan D, Schuring RP, Oskam L, and Richardus JH (2006) Physical distance, genetic relationship, age, and leprosy classification are independent risk factors for leprosy in contacts of patients with leprosy. Journal of Infectious Diseases 193(3): 346–353.
- Rees RJW and Young DB (1994) The microbiology of leprosy. In: Hastings RC (ed.) Leprosy. 2nd edn., pp. 49–83. Edinburgh, UK: Churchill Livingstone.
- Ridley DS and Job CK (1985) The pathology of leprosy. In: Hastings RC (ed.) Leprosy. 2nd edn., pp. 49–83. Edinburgh, UK: Churchill Livingstone.
- van Brakel WH (2000) Peripheral neuropathy in leprosy and its consequences. Leprosy Review 71(supplement): S146–S153.
- van Brakel WH and Anderson AMA (1997) Impairment and disability in leprosy: In search of the missing link. International Journal of Leprosy and Other Mycobacterial Diseases 69(4): 361–376.
- WHO (2005) Global Strategy for Further Reducing the Leprosy Burden and Sustaining Leprosy Control Activities (2006–2010). WHO/CDS/ CPE/CEE/2005.53. Geneva, Switzerland: WHO.
- WHO (2006) Global Strategy for Further Reducing the Leprosy Burden and Sustaining Leprosy Control Activities (2006–2010) – Operational Guidelines. WHO/SEA/GLP/2006.2. New Delhi, India: WHO.
- International Federation of Anti-Leprosy Associations (ILEP) (2001) How to Diagnose and Treat Leprosy. Learning Guide 1. London: ILEP.
- International Leprosy Association (ILA) (2002) Report of the International Leprosy Association Technical Forum, Paris, France, 22–28
- International Journal of Leprosy and Other Mycobacterial Diseases 70(supplement 1): S3–S62.




