Sample Fear Conditioning Research Paper. Browse other research paper examples and check the list of research paper topics for more inspiration. If you need a research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our custom research paper writing service for professional assistance. We offer high-quality assignments for reasonable rates.
1. Basic Terms
The pairing of an initially neutral stimulus (the conditioned stimulus—CS) with a biologically relevant stimulus (the unconditioned stimulus—US) comes to elicit a response (conditioned response—CR) that is usually but not always similar to the response previously associated with the unconditioned stimulus (the unconditioned response—UR). In fear conditioning, the US is an aversive fear-eliciting stimulus such as painful electric shock or loud noise, the CS is a neutral tone or light stimulus. The unconditioned and the conditioned response consist of changes on the subjective, the behavioral and the physiological level and include (in humans) enhanced subjective fear and responses such as freezing, changes in heart rate and skin conductance, the release of stress hormones, reduced pain sensitivity and startle reflex potentiation.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
The development of the CR is based on the formation of an association between a neutral stimulus and a stimulus with innate biological significance (Rescorla 1988). Most studies involving fear conditioning have used cue rather than context conditioning, i.e., discrete CSs were presented rather than using the environment of the animal (e.g., the cage) as CS. In addition, delay conditioning where the CS terminates with the US rather than trace conditioning where the CS and US are separated in time were used in most studies. Fear can be viewed as a specific reaction to threatening stimuli. It can turn into an anxiety disorder when the fear becomes disproportionate to the stimulus that elicits it or when fear is experienced in inappropriate situations.
2. Mechanisms Of Fear Conditioning In Animals
The psychobiological basis of fear conditioning in animals, especially rodents, has been well established (LeDoux 2000, Davis 1992, Maren and Fanselow 1993). When a tone is used as CS and electric shock as US, the auditory CS reaches the lateral nucleus of the amygdala from the thalamus or the cortex, probably depending on the complexity of the CS. When contextual stimuli are employed as CS, the hippocampus is also involved and transmits the signal to the basolateral and the accessory basolateral nucleus of the amygdala. The nociceptive US also reaches the lateral nucleus of the amygdala via thalamic and cortical pathways. Conditioning seems to require an intact lateral nucleus of the amygdala when cue conditioning and an intact basolateral and accessory basolateral nucleus when context conditioning is involved. The central nucleus of the amygdala receives input from the lateral and the basolateral nuclei and it projects to brainstem nuclei that initiate the expression of peripheral fear responses. Whereas the lateral or basolateral nucleus are thus important for the acquisition of the CR, the central nucleus is essential for the expression of the CR (see Figure 1).
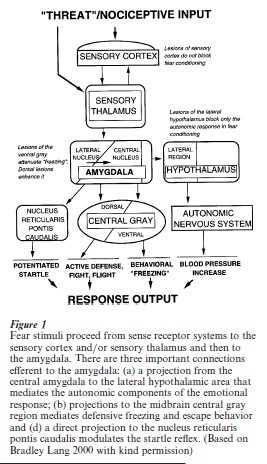
The initial site of plasticity seems to be the lateral nucleus of the amygdala; however, thalamic and cortical reorganization have also been shown during fear conditioning (cf. Weinberger 1998). It is to date not clear if the memory trace is stored in the amygdala or if the amygdala only modulates memories that are stored elsewhere, e.g., in the cerebral cortex. If the amygdala is inactivated during fear conditioning, no CR is formed. If the inactivation occurs after conditioning, then the memory trace is not abolished (LeDoux 2000). In addition, cognitive aspects of fear may involve other pathways.
3. Fear Conditioning In Humans
In humans, the role of the amygdala is less clear. In contrast to rodents, humans possess a much larger frontal cortex and it is likely that it takes over some of the functions that are related to the amygdala in rodents. Damage to the amygdala in humans, has, however, also been related to deficits in fear conditioning whereas lesions in the hippocampus have been shown to interfere with declarative learning and contextual fear conditioning (cf. Bechara et al. 1995). Most studies using positron emission tomography (PET) were unable to replicate the findings from animal experiments that assigned an important role to the amygdala in fear conditioning (for a review see Buchel and Dolan 2000). They instead reported activations in areas such as the hypothalamus, the thalamus, the cingulate gyrus, the premotor and the parietal cortex and, recently, also in the cerebellum (Fischer et al. 2000). Later studies, using differential conditioning paradigms, found some amygdala activation that was positively correlated with activations in the auditory cortex, the frontal cortex and the thalamus. An additional PET study used masked CSs and found significant amygdala activation with the right amygdala showing more activation with unaware (masked) and the left amygdala showing more activation with aware (unmasked) CS presentation. The notion that a direct pathway from the thalamus to the amygdala also exists in humans was recently supported by Morris et al. (1999) who showed that activation of the right amygdala in a masked conditioning paradigm was associated with activity in the superior colliculus and pulvinar but not with activity at cortical sites.
Several recent studies used functional magnetic resonance imaging (f MRI) to study fear conditioning in humans (for a review see Buchel and Dolan 2000). Probably due to the higher spatial and temporal resolution of this method compared to PET, amygdala activation was reported in all studies. In addition to the amygdala, the acquisition of a fear response created activation in the cingulate gyrus and the premotor cortex. Activation of the insulae has also been reported when painful stimuli were used as US. When trace conditioning was employed, activation in the hippocampus in addition to activation in the amygdala and cortical areas was noted.
Based on the observation that not all stimuli are equally easy to condition Seligman (1971) suggested that phylogenetically relevant stimuli such as snakes or spiders might serve as ‘prepared stimuli’ and be more conditionable than others. Subsequently, Ohman (for a summary see Ohman et al. 2000) has shown that conditioned skin conductance responses to a prepared CS such as an angry face or a picture of a snake persist for a longer time (even when the presentation is masked) than when a neutral stimulus such as a picture of a mushroom is used as CS and combined with electric shock as US. Moreover, masked presentations of these stimuli also lead to the acquisition of conditioned responses suggesting that unconscious learning of these stimulus associations is possible.
More recent accounts of fear conditioning have emphasized the role of cognitive variables as well as genetic predisposition in the acquisition of fear (e.g., Davey 1995, Kendler et al. 1992). Part of this may be a cognitive bias termed covariation bias that suggests that aversive outcomes are overestimated in the face of fear-relevant stimuli (e.g., Tomarken et al. 1989). However, although these cognitive variables have recently been discussed extensively as important mediations of the acquisition of fear, the biases themselves can be acquired through conditioning processes and it is not clear to what extent altered cognitive processing precedes or follows the acquisition of fear.
4. Genetic Factors
Genetic factors seem to play a role in the development in anxiety disorders in humans. In familial studies, twin studies and adoption studies show genetic variances of 20 to 40 percent. The same was found for questionnaire variables such as neuroticism. Fear conditioning ability and predisposition to learn fear responses were extensively studied in animals. In rodents, fear processes or anxiety measured in the open field test or avoidance conditioning have revealed a strong genetic basis explaining most of the interindividual variance. However, the genes responsible for that trait have not yet been identified.
The situation is different for antisocial personality disorder and criminal behavior. Assuming that a deficit in anticipatory fear conditioning is the central symptom from which all consequent social problems follow, all evidence ranging from twin to adoption studies and gene sequencing suggests a moderate to strong genetic influence. If sensitivity to alcoholism and novelty-seeking are included as comorbidity factors the genetic variance increases further. Whether this is related to genetic fear conditioning remains to be seen (Plomin et al. 2001).
5. Significance Of Fear Conditioning For The Understanding Of Anxiety Disorders
Although classical fear conditioning has been implicated as an important mechanism in the development of most anxiety disorders, studies that support the etiological significance of fear conditioning have been rare. Retrospective data from either adult or child patients or the parents of children with specific phobias revealed that about 40–75 percent of the subjects or the parents could recall a traumatic event (e.g., Menzies and Clarke 1993), a substantial number of the remaining subjects experienced vicarious conditioning or had been verbally informed about potential dangers related to the feared object. In evaluating these data it must be kept in mind that all these reports are retrospective and that fear conditioning may be nondeclarative and therefore not be accessible to conscious recollection. Of greater relevance are therefore studies that examine fear conditioning in clinical groups or analogue samples.
In social phobics, several studies used differential conditioning with faces as CS and aversive odor or shock as US. In two studies, a conditioned response was already present prior to the acquisition phase with the social phobics showing enhanced amygdala and (in one study) orbitofrontal responses to the face CS suggesting that they displayed a fear response to neutral faces that never elicits amygdala activation in healthy humans (e.g., Birbaumer et al. 2000). In another study stronger amygdala activation was present to the CS+ than the CS+ only in social phobics whereas the healthy controls showed deactivation. However, the scanning was performed late in the acquisition phase and it has been shown that the amygdala is only responsive early in the acquisition process. Additional studies that used conditioning paradigms or phobia-relevant cues during imaging also found activation increases in the anterior cingulate and the insulae in accordance with the findings of fear conditioning in healthy subjects.
In persons with psychopathy and antisocial personality disorder disturbed fear conditioning seems to be present with a concomitant complete absence of orbitofrontal, insular and cingulate activation during fear conditioning (Birbaumer et al. 2000). This absence of associative learning with respect to the aversive consequences of a stimulus might be the basis of the antisocial and often criminal behavior of these persons.
In posttraumatic stress disorder, higher conditionability as evident in enhanced and more difficult to extinguish fear responses has been observed. It is, however, not clear to what extent this condition predated the disorder or if it must be viewed as a consequence of the disorder and to what extent conditioning of trauma-related stimuli actually occurred because none of the studies involved trauma relevant cues (e.g., Orr et al. 2000).
6. Future Perspectives
Future research must focus on the elucidation of the neuronal correlates of fear conditioning in healthy humans and patients. It is not clear to what extent the amygdala plays the same role in fear conditioning in humans as it does in rodents. Furthermore, the role of awareness of the CS and the CS–US relationship has not been determined by neuroimaging methods. The importance of fear conditioning for the understanding of anxiety disorders needs to be explored using paradigms that involve fear-relevant and prepared stimuli as well as longitudinal measurements. Finally, the role of cognitive and genetic variables and their relationship to fear conditioning needs to be further explored.
Bibliography:
- Buchel C, Dolan R J 2000 Classical fear conditioning in functional neuroimaging. Current Opinion in Neurology 10: 219–23
- Bechara A, Tranel D, Damasio H, Adophs R, Rockland C, Damasio A R 1995 Double dissociation of conditioning and declarative knowledge relative to the amygdala and the hippocampus in humans. Science 269: 1115–18
- Birbaumer N, Veit R, Flor H, Patrick C 2000 Excess and lack of fear: an f MRI study with social phobias and psychopaths. Society for Neuroscience Abstracts 26: S1072
- Bradley M M, Lang P J 2000 Measuring emotion: behavior, feeling and physiology. In: Lane R D, Nadel L (eds.) Cognitive Neuroscience of Emotion. Oxford University Press, New York, pp. 242–76
- Davey G C 1995 UCS revaluation and conditioning models of acquired fears. Behavior Research and Therapy 27: 521–8
- Davis M 1992 The role of the amygdala in fear and anxiety. Annual Review of Neuroscience 15: 353–75
- Fischer H, Andersson J L, Furmark T, Fredrikson M 2000 Fear conditioning and brain activity: a positron emission tomography study in humans. Behavioral Neuroscience 114: 671–80
- Kendler K S, Neale M C, Kessler R C, Heath A C, Eaves L J 1992 The genetic epidemiology of phobias in women: the interrelationship of agoraphobia, social phobia, situational phobia and simple phobia. Archives of General Psychiatry 49: 273–81
- LeDoux J E 2000 Emotion circuits in the brain. Annual Review of Neuroscience 23: 155–84
- Maren S, Fanselow M S 1993 The amygdala and fear conditioning: has the nut been cracked? Neuron 16: 237–40
- Menzies R G, Clarke J C 1993 The etiology of childhood water phobia. Behavior Research and Therapy 31: 499–501
- Morris J S, Ohman A, Dolan R J 1999 A subcortical pathway to the right amygdala mediating ‘unseen’ fear. Proceedings of the National Academy of Science USA 96: 1680–5
- Ohman A, Flykt A, Lundqvist D 2000 Unconscious emotion: evolutionary perspectives, psychophysiological data and neuropsychological mechanisms. In: Lane R D, Nadel L (eds.) Cognitive Neuroscience of Emotion. Oxford University Press, Oxford, pp. 296–327
- Orr S P, Metzger L J, Lasko N B, Macklin M L, Peri T, Pitman R K 2000 De novo conditioning in trauma-exposed individuals with and without posttraumatic stress disorder. Journal of Abnormal Psychology 109: 290–8
- Plomin R, DeFries J, McClearen G, Roth M 2001 Behavioral Genetics 4th Edn. Freeman, San Francisco, CA
- Rescorla R A 1988 Pavlovian conditioning: It’s not what you think it is. American Psychologist 43: 151–60
- Seligman M E P 1971 Phobias and preparedness. Behavior Therapy 2: 307–20
- Tomarken A J, Mineka S, Cook M 1989 Fear-relevant selective associations and covariation bias. Journal of Abnormal Psychology 98: 381–94
- Weinberger N M 1998 Physiological memory in primary audi tory cortex: characteristics and mechanisms. Neurobiology of Learning and Memory 70: 226–51




