View sample cancer research paper on environmental carcinogens. Browse other research paper examples for more inspiration. If you need a thorough research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Definition And History
This research paper defines both environmental and occupational causes of cancer as either chemical or physical agents in the workplace or in the general environment to which individuals are involuntarily exposed. Although most sources consistently define occupational causes of cancer as involuntary exposure to agents found in the workplace, the definition of environmental causes of cancer varies and can include lifestyle factors (such as smoking and alcohol consumption), reproductive factors, diet and infectious agents.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
Much of the scientific evidence linking cancer with involuntary occupational and environmental agents comes from studies of workers who often experience more intense and prolonged exposures than the general public. One of the earliest examples of cancer caused by involuntary exposures is that of Percival Pott’s 1775 observation of an excess incidence of scrotal cancer among young chimney sweeps in London. Studies later confirmed Pott’s observation and identified polycyclic aromatic hydrocarbons (PAHs), particularly benzo-(a)-pyrene, as the causal agent.
The Debate: How Much Cancer Is Caused By Involuntary Occupational And Environmental Exposures?
The discussion regarding the contribution of environmental and occupational exposures to the overall cancer burden has been an area of contention for at least the past three decades, since the assertion in 1977 by Higginson and Muir that 80% of all cancers were due to environmental exposures. The evidence that Higginson and Muir invoked in their seminal article included, ‘‘descriptive epidemiological data relating to migrants, geographical variation in incidence, changes in risk over time, correlation studies, clusters and case reports’’ (Higginson and Muir, 1977). Although these authors were referring to ‘‘widespread general exposures of air and water pollution, the work environment, exposures resulting from personal choice such as smoking and drinking, and the diet,’’ the concern that involuntary exposures to substances in the air, water, and work environment are major contributors to cancer in humans has persisted.
In the past three decades, there have been several efforts to estimate the proportion of cancer due to these involuntary exposures, starting with an ambitious effort by Doll and Peto (1981) and more recently by a group of authors at the Harvard Center for Cancer Prevention (1996). These well-cited sources have estimated that roughly 2% of cancer deaths are due to pollution and 4% to occupation. However, some (including Doll and Peto themselves) have argued that assigning categories of exposure-specific percentages in causing cancer that total 100% is erroneous, given what we know about how cancer is caused (Clapp et al., 2006). Current knowledge of the mechanisms of cancer suggests that all cancers evolve from a complicated combination of multiple exposures. One recent description of this dynamic process reduces it to six essential alternations that may overwhelm the natural defenses built into human cells and tissues to produce a tumor (Hanahan and Weinberg, 2000). Assuming that this mechanism is correct, no one exposure singlehandedly causes cancer; all categories of cancer causes (genetic, environmental, occupational, behavioral/lifestyle, etc.) are likely to play a role, to some extent, in the development of an individual’s cancer. Consequently, the number of cancer cases in which environmental and occupational factors play a role in the development of the disease is likely to be extremely high, given the ubiquitous presence of some known carcinogens.
Cancer Clusters
People occasionally perceive clusters of cancer in their communities or workplaces and believe that they must have been caused by a common environmental exposure. These concerns are understandable and often lead to some type of investigation or study to determine the cause. Often, there are small numbers or rare types of cancers involved. This is one of the most vexing issues facing occupational and environmental health because tools to investigate cancer clusters are crude and often inadequate. Statistical methods to evaluate cancer clusters typically lack power to detect a significant excess, even if it is there in a community or workplace.
Cancer clusters can and do occur because of exposures from a common source. There are several famous examples of this, including the cluster of angiosarcoma of the liver in workers exposed to vinyl chloride at a manufacturing plant, the cluster of clear-cell adenocarcinoma of the vagina in offspring of women who took DES, and the cluster of childhood leukemia in Woburn, Massachusetts residents exposed to contaminated drinking water. These examples give validity to concerns that exposures in other communities or workplaces might also generate legitimate cancer clusters, although it may be difficult or even impossible to determine this with presently available tools. History has shown that some clusters are indeed signals that a preventable exposure occurred, but exposures linked to perceived clusters can be difficult to document. The proper response to such health concerns is not to dismiss them as improbable statistical artifacts but to engage concerned families or workers and public health representatives in honest communication about what is known and what is not known about the exposures and the cancers that are perceived to constitute a cluster.
Environmental And Occupational Carcinogens: The State Of The Science
The following sections provide a summary by cancer site of the state of the science relating to environmental and occupational exposures that are known or suspected to cause cancer. Only those cancer types that have substantial literature describing established and suspected causal associations with environmental and occupational agents in humans are described, and we refer readers to other reviews detailing other cancer types (see Bibliography: section). Although scientific evidence regarding causes of cancer is based on several disciplines of study, the evidence provided in this review is primarily from epidemiologic studies of groups of individuals exposed to agents at work or in their communities. Much of the experimental evidence of carcinogenicity comes from animal studies that would not be possible or ethical in humans. When direct human data are not available, it is both necessary and appropriate to extrapolate from animal studies. At present, there are many more chemicals that have been shown to cause cancer in experimental animals than are considered established human carcinogens, although the number for which sufficient human data exist is increasing. This research paper does not address the complexities of timing of exposure, dose, and additive or synergistic effects of multiple exposures, but a rapidly growing body of evidence points to their importance (see Bibliography: section). Similarly, this research paper does not address racial and socioeconomic disparities in cancer risk and differential exposures to occupational and environmental carcinogens (see Bibliography: section).
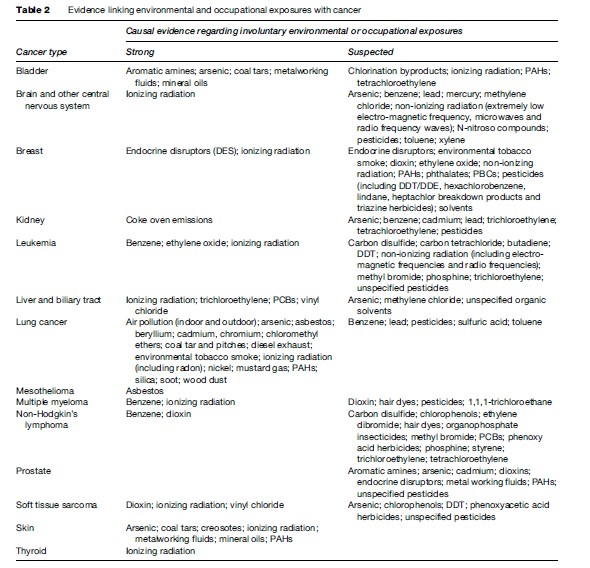
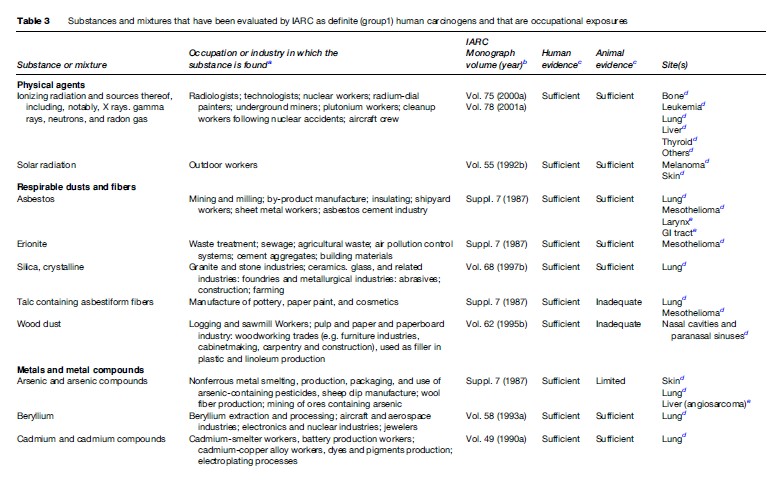
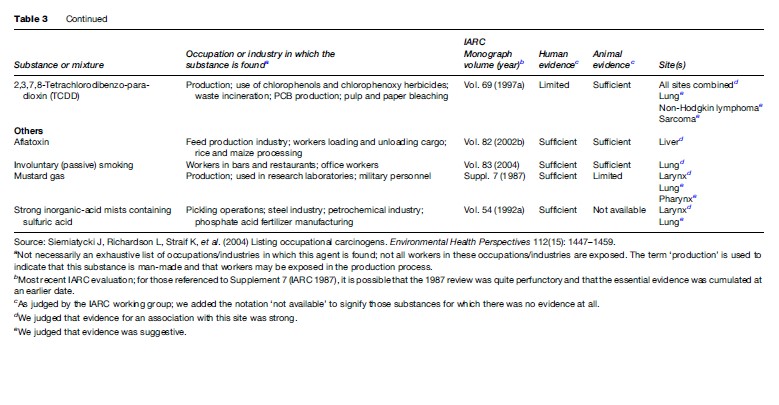
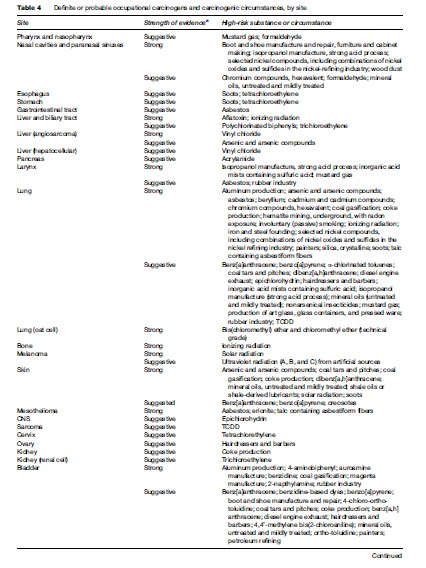
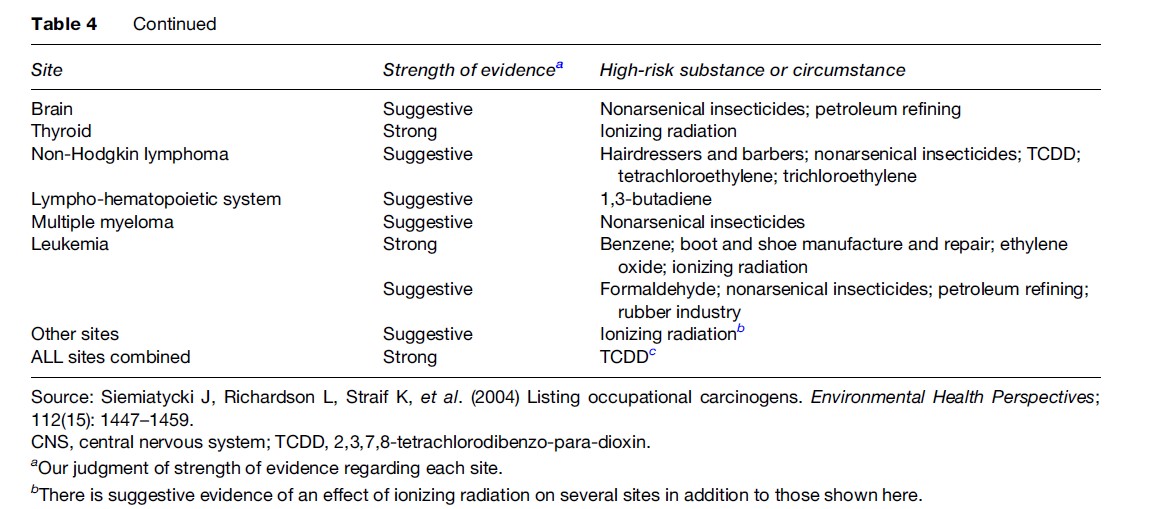
Table 1 briefly outlines the sources and uses of most of the carcinogenic agents reviewed. A brief overview of the causes of cancer reviewed in this research paper can be found in Table 2. See Tables 3–4, developed by Siemiatycki et al. (2004), for additional information on substances and industries classified as definite (group 1) carcinogens by the International Agency for Research on Cancer (IARC), occupational exposures to them, and the cancer sites with which they are associated. From 1972 through 2003, the International Agency for Research on Cancer (IARC) evaluated over 880 substances, complex mixtures, and industrial processes. IARC classified 89 of these substances as definite human carcinogens, 64 as probable human carcinogens, and 264 as possible human carcinogens. Siemiatycki et al. (2004) determined that these groupings consisted of 28 definite, 27 probable, and 113 possible human occupational carcinogens and identified 18 occupations or industries that have been considered by IARC to definitely, probably, or possibly entail ‘‘excess risk of cancer among workers’’ (Siemiatycki et al., 2004).
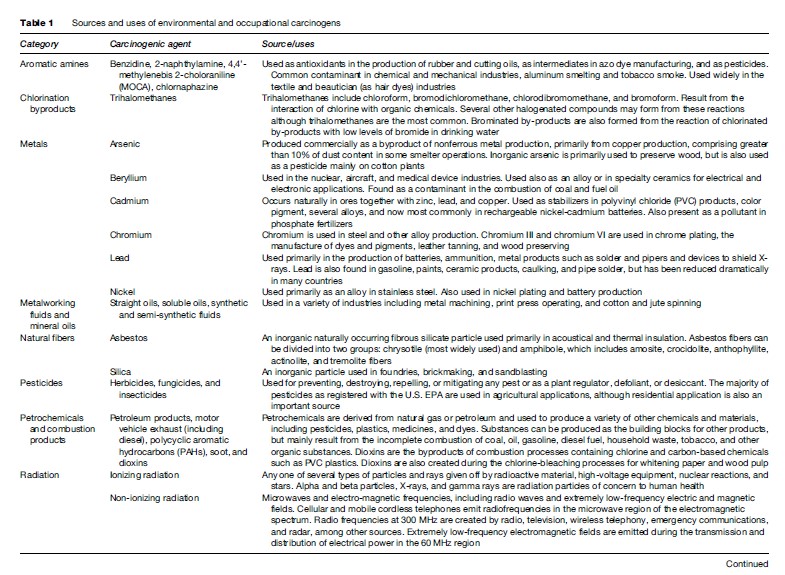
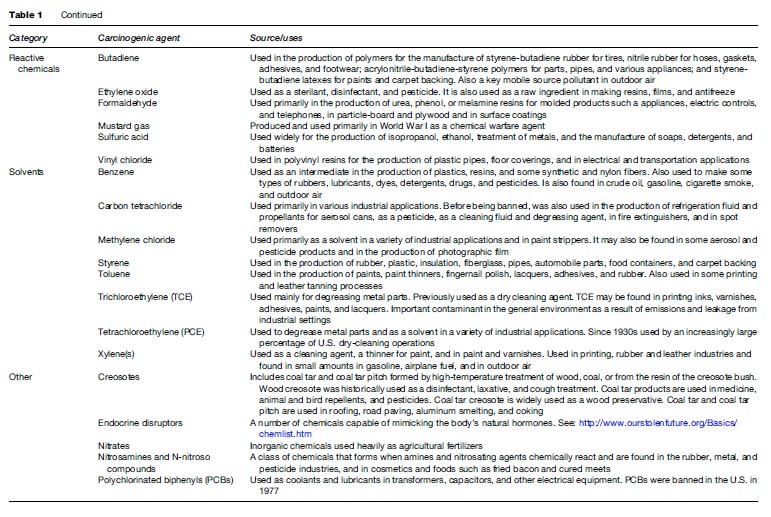
Bladder Cancer
The epidemiologic evidence linking metal exposure, specifically arsenic, with bladder cancer is strong and extensive. Much of the evidence comes from epidemiologic studies conducted in regions with high concentrations of inorganic arsenic contaminants in drinking water and in medicinal formulations such as Fowler’s solution. A Belgian case-control study suggests cadmium may also be a bladder carcinogen based on a nearly six-fold increased risk among those with the highest blood levels of cadmium; however, further confirmation of this association is needed. Several volatile chemicals have been linked with bladder cancer. Evidence from multiple studies examining chlorination by-products has consistently found elevated risk of bladder cancer, especially among populations with long-term exposure to chlorinated water. Risk of bladder cancer from exposure to solvents is also suspected, particularly for the solvent tetrachloroethylene (PCE), as documented in studies of dry cleaning workers.
Aromatic amines (arylamines), including 2-naphthylamine (b-naphthylamine), benzidine, 4-aminobiphenyl, and chlornaphazine (a derivative of 2-naphthylamine previously used in the treatment of polycythemia), as well as the manufacturing of auramine and magenta dye, are well-established causes of bladder cancer and one of the first carcinogens to be associated with an occupational exposure. Studies of several other aromatic amines, including O-toluidine and aniline, have demonstrated elevated risks associated with bladder cancer. Strong evidence demonstrates that workers in the rubber industry are at elevated risk for bladder cancer. Elevated risk of bladder cancer has also been observed among occupations exposed to hair dyes.
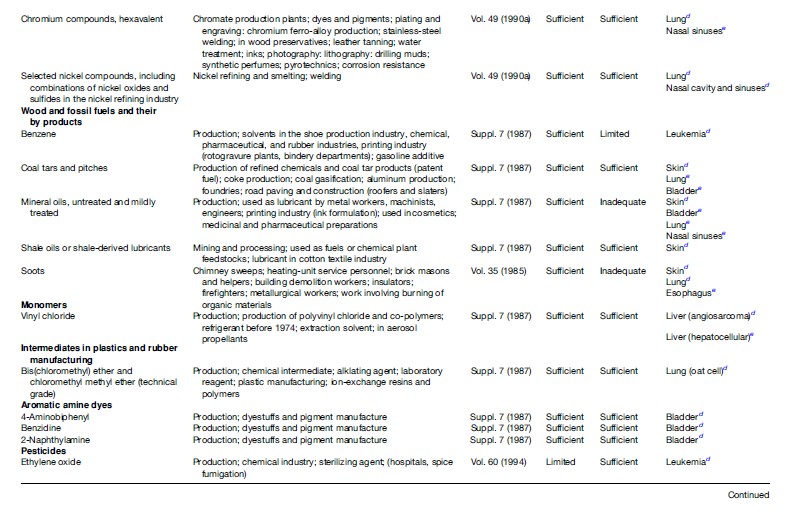
A number of epidemiologic studies have documented an increased risk of bladder cancer among workers exposed to petrochemicals and combustion products in different industries, thus suggesting an association with polycyclic aromatic hydrocarbons (PAHs), their nitroderivatives, and to diesel exhausts. An increase of bladder cancer risk, although inconsistent, is also found among industries with high exposure to PAHs from coal tars and pitches. Studies of workers using metalworking fluids and mineral oils offer strong evidence for an association with bladder cancer. Recent reviews of studies of A-bomb survivors have documented elevated risks of bladder cancer associated with ionizing radiation. Other agents possibly associated with bladder cancer are seen in occupations entailing exposures to leather dusts, solvents other than PCE, paints and inks, and coal tar and pitches.
Brain And Other Central Nervous System Cancers
Cancer of the brain and the central nervous system (CNS) is relatively rare in adults but is one of the most common cancers in children.
Metals, primarily exposure to lead, have been weakly supported as risk factors of brain cancer by several studies, including a meta-analysis of eight studies of populations with high occupational exposures to lead. Additional studies provide limited evidence for increased risk of brain or CNS cancers and exposure to arsenic and mercury. Studies have suggested an association between exposure to solvents including benzene, toluene, xylene, and methylene chloride (particularly among women) and brain cancer. Studies of fathers occupationally exposed to solvents as well as paints and/or inks provide limited evidence for increased risk of brain or CNS cancers among their children.
Ionizing radiation is a proven etiologic agent associated with brain cancer based on evidence from therapeutic radiation studies and children exposed to diagnostic radiation in utero. The evidence regarding risk of brain cancer from exposure to non-ionizing radiation from extremely low-frequency electromagnetic fields is considered strongly suggestive, based on studies examining both workers and children. However, paternal exposure to electromagnetic fields associated with elevations of childhood nervous system cancers has also been suggested. Studies are conflicting regarding the risk of brain cancer from exposure to microwaves and radio frequencies, primarily from cellular phone use, and exposure to radio and TV transmitters and are limited by poor detail on actual exposures, short follow-up periods, and a lack of consistency regarding tumor grade and location.
Numerous studies have demonstrated that pesticide exposure is associated with CNS and brain cancer among children and adults. Studies generally found greater risks among children associated with parental exposure to pesticides prior to conception and during pregnancy than for exposures experienced during childhood.
Multiple studies examining frequent maternal consumption of cured meats during pregnancy indicate that exposure to N-nitroso compounds increases the risk of CNS tumors in children. Scientists have found some evidence for increases of brain and CNS cancers among women in various industries, including laboratories, rubber, painting, plastics, metals, wool and textile spinning, and petroleum refining.
Breast Cancer
Breast cancer is by far the most commonly diagnosed cancer in women in industrialized countries.
The etiology of breast cancer may be among the most complicated of all cancers given inherent, life-long exposures to multiple endogenous and exogenous factors. Timing and dose are likely to have particular relevance to the developing bodies of girls.
Ionizing radiation is the best and longest established exogenous environmental cause of breast cancer based on analyses of A-bomb survivors and medical radiation studies. Additional possible links to breast cancer have been established for non-ionizing radiation from electromagnetic fields (EMFs).
Endocrine disruptors (also known as xenoestrogens and synthetic estrogens) mimic the actions of estrogens and are found in many pesticides, fuels, plastics, detergents, and prescription drugs. The tragic story of DES (diethylstilbestrol) has provided some of the most convincing evidence that synthetic chemicals can act like hormones. Daughters of women who took DES during pregnancy have more than twice the breast cancer risk of women in their age brackets who were not exposed to DES in utero.
Unspecified solvents have been linked to increased breast cancer risk, particularly in occupational settings such as electronics, semiconductor industries, metal fabrication, lumber, furniture production, printing, chemical, and textile and clothing industries. Styrene, benzene, carbon tetrachloride, and formaldehyde are some of the specific solvents suspected to increase breast cancer risk.
The research is not clear with regard to breast cancer and pesticide exposure. Some studies have established probable links with a number of pesticides, including dichloro-diphenyl-trichloroethane/dichlorodiphenyldichloroethylene (DDT/DDE), hexachlorobenzene (HCB), hexachlorocyclohexane (lindane), heptachlor epoxide (a breakdown product of the insecticide heptachlor), and triazine herbicides (including atrazine). However, well-designed cohort studies, such as the U.S. Agricultural Health Study – whose preliminary results reveal significantly increased risk of breast cancer among women whose husbands used a variety of pesticides – will help clarify such links.
The sum of the literature examining links between breast cancer and polychlorinated biphenyls (PCBs) is vast and inconclusive. However, a synthesis of this vast literature noted that women with a polymorphism in the CYP1A1 gene were at greater breast cancer risk when exposed to PCBs.
Several lines of evidence suggest that environmental tobacco smoke (ETS) – a complex mixture of nearly 5000 chemical compounds, 43 of which are known human or animal carcinogens – is a likely cause of breast cancer among pre-menopausal women. Combustion byproducts, specifically PAHs, are one of the components of environmental tobacco smoke. Among premenopausal women in both occupational settings and the general environment, PAHs are suspected to increase breast cancer risk. Concern for the role of PAHs in breast cancer development has more recently been extended to male breast cancer as well. Other combustion byproducts such as dioxin are suspected to increase breast cancer, as well as reactive chemicals, including ethylene oxide.
Kidney Cancer
Cancer of the kidney and renal pelvis is less common than bladder cancer, with which it shares some common risk factors. Kidney cancer has been linked to exposure to some metals, including arsenic, cadmium, and lead. Although not considered conclusive, several studies of arsenic exposure in drinking water in regions of South America and Taiwan have documented excess mortality from kidney cancer. Studies examining kidney cancer in relation to lead exposure provide some evidence (albeit weak) of a causal link.
Links have also been established with kidney cancer and solvent exposure. A thorough review of over 80 published papers and letters examining cancer epidemiology associated with exposure to trichloroethylene (TCE) found strong and consistent evidence of an increased risk of kidney cancer, primarily based on studies that assessed exposure using urinary biomarkers (Wartenberg et al., 2000). The review similarly found some evidence to suspect elevated rates of kidney cancer among laundry and dry cleaning workers exposed to PCE. Increased kidney cancer rates have also been observed among workers exposed to gasoline, particularly those who distribute gasoline.
Several studies demonstrate an association with Wilm’s tumor (a childhood cancer of the kidney) and exposure to pesticides (Zahm and Ward, 1998). Paternal employment as welder or mechanic has also been suggested as a risk factor for Wilm’s tumor in children.
Leukemia
Leukemia has been relatively rare in adults but is the most common cancer in children, and childhood leukemia rates have been rising for the past few decades.
Workers exposed to organic solvents have shown significantly elevated mortality from leukemia. Benzene is etiologically related to the development of leukemia, specifically acute non-lymphocytic leukemia. A large-scale cohort study in China (a collaboration of the NCI and the Chinese Academy of Preventive Medicine) has linked benzene to other leukemia subtypes even at low levels of exposure (Hayes et al., 2001). Evidence for an association between childhood leukemia and paternal exposure to solvents, including benzene, carbon tetrachloride, and TCE, as well as to paints and pigments, is also quite strong. Strong evidence demonstrates that employment in the rubber industry entails an elevated risk for leukemia, probably due to exposure to benzene, other solvents, and reactive chemicals, such as 1,3-butadiene. There is strong evidence for increased risk of leukemia from exposure to other reactive chemicals, such as ethylene oxide.
Exposure to ionizing radiation is a well-recognized cause of leukemia. Prenatal exposure to diagnostic radiography of mothers during pregnancy is an established cause of childhood leukemia. The evidence is conflicting regarding the risk of leukemia from exposure to nonionizing radiation, including electromagnetic frequencies (EMFs). However, on balance, a precautionary approach regarding exposure to EMFs is warranted, particularly for childhood leukemia.
Substantial evidence indicates that exposure to pesticides increases the risk of leukemia in both adults and children. Over a dozen studies found elevated rates of leukemia among children whose parents were occupationally exposed to pesticides or who used pesticides in their home or garden. Occupational studies of workers exposed to pesticides consistently demonstrate increased risk and mortality, and evidence regarding risk associated with specific pesticides will be more firmly established as data emerge from studies such as the U.S. Agricultural Health Study.
Liver And Biliary Cancer
The incidence of liver and biliary cancer is low in industrialized nations, although it has been rising in recent years. This cancer is one of the most common in the developing nations of Africa and Asia. The major risk factors for liver cancer in these countries are aflatoxin and the hepatitis virus.
Liver cancer has been linked with exposure to metals, primarily arsenic. Although not considered definitive, several studies suggest that ingesting arsenic in drinking water is associated with liver cancer.
Strong evidence supports significantly elevated mortality from cancer of the liver and biliary tract among workers exposed to organic solvents. Specific solvents, including TCE and methylene chloride, are suspected to increase the risk of developing liver and biliary cancers.
Exposure to ionizing radiation is a well-established cause of liver cancer. Some evidence is offered for elevated risk of liver cancer associated with reactive chemicals. Vinyl chloride-exposed populations have an elevated rate of mortality from angiosarcoma of the liver. An additional strong risk factor for liver cancer includes PCBs.
Lung Cancer
Lung cancer is the second most commonly diagnosed cancer in much of the world, and it is usually fatal. Recent trends in industrialized countries indicate a reduction in male lung cancer incidence and mortality, and female lung cancer may follow this pattern in coming years.
Exposure to a number of metals has been linked to an increased risk of lung cancer. Strong evidence from multiple studies has demonstrated increased risk of mortality due to lung cancer from exposure to arsenic dusts. Increased risk of lung cancer has also been observed among workers involved in the manufacturing of arsenical pesticides. Studies of arsenic contamination in drinking water as a result of either natural or industrial contamination have consistently demonstrated increased risks for lung cancer. Beryllium exposure among workers is considered an established cause, as is exposure to cadmium and chromium (primarily hexavalent chromium salts), based on evidence from occupational studies. Studies in workers show that some nickel compounds (sparingly soluble and soluble) are linked to lung cancer.
Exposure to ionizing radiation is a well-recognized cause of lung cancer. In addition to studies of survivors of the atomic bomb, radon-exposed miners and individuals exposed to radon in their homes have been consistently linked to elevated risk of lung cancer.
Workers exposed to reactive chemicals have elevated risk of lung cancer. Bis (chloromethyl) ether (BCME) and chloromethyl methyl ether (CMME), used primarily in the preparation of anion exchange resins, are established occupational carcinogens of the lung. Exposure to mustard gas is also a well-known cause of lung cancer. Sulfuric acid is a suspected cause of lung cancer among exposed workers.
Exposure to environmental tobacco smoke (ETS) is an established cause of lung cancer based on numerous studies, although the number of cases caused by direct or mainstream tobacco smoke is much greater.
Studies of varied designs and diverse settings have repeatedly found rates of lung cancer associated with outdoor air pollution, mainly from exposure to fossil fuel vapors. Although examination of carcinogenic risks from individual chemicals in air pollution is difficult, there is a biological rationale for links to cancer from numerous compounds including benzo[a]pyrene, benzene, some metals, particles, and possibly ozone. Studies in regions of China and other countries of indoor air pollution from combustion sources used for heating and cooking, as well as high levels of cooking oil vapors, have identified these exposures as risk factors for lung cancer.
Substantial evidence examining both occupational and residential exposure to petrochemicals and combustion byproducts provides support for an association with lung cancer. Exposure to PAHs has been repeatedly shown to increase lung cancer risk. Suggestive evidence supports a causal link between lung cancer and exposure to coal tar and pitches, and strong evidence supports a link to soot. Evidence from populations most highly exposed to dioxin provides support for an increased risk of lung cancer.
Elevations of lung cancer have been observed in occupational studies examining exposure to pesticides, notably DDT, although these findings are somewhat inconsistent. More recently, some evidence has emerged relating to an increased risk of lung cancer associated with the insecticides chlorpyrifos and diazinon and the herbicides metolachlor and pendimethalin, especially among pesticide applicators. Several studies of printing workers exposed to metalworking fluids based on mineral oil formulations have found excess lung cancer. Strong evidence supports an increased risk of lung cancer associated with exposure to silica and natural fibers, including asbestos (all fiber types) and other mineral fibers, although evidence is conflicting for man-made fibers such as glass wool, rock/slag wool, and ceramic fibers. Some evidence supports an excess risk of lung cancer in other specific industries, including the rubber industry.
Mesothelioma
Mesothelioma is a rare but extremely fatal cancer. Exposure to the natural fiber asbestos (all fiber types) is an established cause of mesothelioma of the pleura and peritoneum.
Multiple Myeloma
Multiple myeloma is somewhat rarer than leukemia in adults, and it shares many of the same occupational and environmental risk factors with the hematopoietic and lymphatic system malignancies.
Solvent (particularly benzene) and ionizing radiation exposure has been linked to increased risk of multiple myeloma. Exposure to various pesticides, including those contaminated with dioxin, has been associated with multiple myeloma in some studies. Limited evidence suggests an elevated risk of multiple myeloma associated with personal and occupational exposure to hair dyes.
Non-Hodgkin’s Lymphoma
Non-Hodgkin’s lymphoma (NHL) is the most common cancer of the hematopoietic and lymphatic system.
Strong evidence supports an increased risk of NHL following occupational exposure to organic solvents, particularly benzene exposure. Benzene is also suspected in increases in NHL observed among children living near railways, oil refineries, and petrochemical plants. There is also support for increased risk of NHL following exposure to TCE, PCE, and styrene.
Although the evidence is somewhat conflicting, multiple studies have documented elevated risks of NHL among agricultural and forestry workers exposed to pesticides. Of studies that have examined specific pesticides, increased risk and death from NHL have been associated with phenoxy acid herbicides, chlorophenols and organophosphorous insecticides, carbon disulfide, phosphine, methyl bromide, and ethylene dibromide. Limited evidence from a number of studies of occupational exposures to DDT and a case-control study examining adipose tissue levels of other organochlorine pesticides (i.e., dieldrin, oxychlordane, heptachlor) provides some support for increased risks of NHL. Evidence from a few studies provides limited support for an increased risk of childhood lymphoma (including both Hodgkin’s lymphoma and NHL) associated with parental occupational exposure to pesticides.
Substantial evidence links NHL with dioxin exposure, although not all studies are in agreement. Several studies have linked higher chlorinated congeners of PCBs in adipose tissue with NHL, consistent with findings that PCBs are immunotoxic substances. A review of the epidemiologic evidence regarding occupational and personal exposure to hair dyes suggests that hair dye exposure can produce a small elevation in NHL risk.
Prostate Cancer
Prostate cancer is by far the most commonly diagnosed cancer in men and is the second leading cause of cancer death in men. Studies of the extreme geographic variations in prostate cancer incidence around the globe (up to 30-fold) and changes in incidence or mortality among migrants from developing to developed countries provide the strongest evidence that diet and/or environmental factors play a significant role in prostate cancer etiology.
Cooking red meat produces various aromatic amines, many of which have proven to be carcinogens in animal studies. One of the aromatic amines (2-amino-1-methyl-6-phenylimidazol[4,5-b]pyridine) is known to cause invasive prostate cancer in rats.
Metals, including cadmium and arsenic, have been linked to prostate cancer. Studies also suggest increased risk of prostate cancer associated with pesticide exposures, notably herbicides. A number of occupational studies, though not all, have shown an elevated risk of prostate cancer incidence and/or mortality among farmers and pesticide applicators. The findings of elevated risk for the latter group are particularly significant.
Considerable evidence associates exposure to endocrinedisrupting chemicals in the environment with prostate cancer and suggests that the timing of the exposure is critical, especially in the cases of fetuses and developing boys.
Excess risks have also been found for exposure to metalworking fluids, and liquid fuel combustion products, dioxin, and other substances.
Soft Tissue Sarcomas (STS)
Soft tissue sarcomas (STS) (including heart) are rare cancers of the muscle, fat, and connective tissue.
Evidence supporting a link between soft tissue sarcomas and metals is provided only in some studies suggesting elevated rates of STS among patients treated with arsenical medications. The reactive chemical vinyl chloride monomer is an established cause of angiosarcoma of the liver. There is suggestive evidence of an association between occupational arsenic exposure and angiosarcoma of the liver. Exposure to ionizing radiation among radium workers and patients receiving medical treatment is a recognized cause of both bone and sinus sarcomas but is less strongly recognized in association with STS.
Risk of STS has repeatedly been elevated in studies examining exposure to pesticides among farmers, forestry and horticultural workers, and pesticide applicators.
Strong evidence supports excess risk of STS associated with exposure to dioxin. Limited evidence from one case control study suggests that fluoridated water could increase risk of osteosarcoma.
Skin Cancer
Non-melanoma skin cancers (namely basal and squamous cell) are by far the most commonly diagnosed cancers, though they are rarely fatal. Melanoma of the skin is relatively rare among Blacks, but among Whites, the incidence rate has been increasing rapidly in many parts of the world in recent years.
Ionizing radiation is a well-recognized cause of nonmelanoma skin cancer based on evidence from studies of atomic bomb survivors and radiologists. Exposure to ultraviolet radiation and sun exposure is a definitive cause of all types of skin cancer including melanoma.
Metals such as inorganic arsenic are well recognized as skin carcinogens. Studies examining medicinal uses of arsenic, such as Fowler’s solution (potassium arsenic), also demonstrated skin carcinogenicity. There is established evidence that early formulations of metalworking fluids and mineral oils used in cotton and jute spinning and metal machining were carcinogenic to the skin and an established cause of scrotal cancer among workers in the textile and metalworking industries.
Exposure to creosotes used as wood preservatives is an established cause of non-melanoma skin cancer in addition to exposure to PAHs and coal tars. In the 1950s, researchers identified an increased incidence of scrotal cancer among men working with cutting oils.
Among other specific occupations, limited evidence supports an increased risk of skin cancer, particularly melanoma, among workers in the petroleum industry and fire-fighters.
Thyroid Cancer
Thyroid cancer incidence has increased in recent years, although this rare cancer is seldom fatal. Survivors of the atomic bomb, patients treated medically with radiation, workers at nuclear facilities, and residents exposed to radioactive fall-out are all part of the body of evidence definitively linking exposure to ionizing radiation as a cause of thyroid cancer. Recent evidence from studies of the Chernobyl accident demonstrates strong concern for increased risk of thyroid cancer among exposed children.
Conclusions
The scientific literature provides substantial evidence of involuntary environmental and occupational causes of cancer and fully justifies ongoing efforts to prevent carcinogenic exposures.
Even though tobacco smoke remains the single most significant preventable cause of cancer, it has been linked neither to the majority of cancers nor to many of the cancers that have increased rapidly in recent decades, including melanoma, lymphomas, testicular, brain, and bone marrow cancers. The rise and fall of lung cancer has tracked the rise and fall of the prevalence of smoking, with expected, distinct time delays for men and for women.
If ethnicity were to play a significant role in determining cancer risk, then immigrants should retain the cancer incidence rates of their country of origin. Yet immigrants to a new land acquire the cancer rates of their new home within one to two generations.
Elevated cancer rates follow additional patterns – the disease is more common in cities, in farming states, near hazardous waste sites, downwind of certain industrial activities, and around certain drinking-water wells. Patterns of elevated cancer incidence and mortality have been linked to areas of pesticide use, toxic work exposures, hazardous waste incinerators, and other sources of pollution.
Farmers in industrialized nations die more often than the rest of the population from multiple myeloma, melanoma, prostate cancer, Hodgkin’s lymphoma, leukemia, and cancers of the lip and stomach. They have higher rates of non-Hodgkin’s lymphoma and brain cancer.
A considerable portion of the evidence presented above derives from occupational studies, in part because the workplace can provide the structure that epidemiologic studies need. Unequal workplace exposures among different populations provide further indications of the ability of occupational exposures to cause harm.
In this research paper, we have briefly summarized the evidence of some of the most compelling involuntary environmental and occupational links to cancer. This summary does not encompass the full body of scientific knowledge, including the rapidly developing genetic and mechanistic understandings of how cancer arises. It is nevertheless clear that there are substantial preventable exposures to occupational and environmental carcinogens that justify ongoing efforts to reduce their contribution to the cancer burden.
Bibliography:
- Clapp RW, Howe GK, and Jacobs M (2006) Environmental and occupational causes of cancer re-visited. Journal of Public Health Policy 27(1): 61–76.
- Doll R and Peto R (1981) The causes of cancer: Quantitative estimates of avoidable risks of cancer in the United States today. Journal of the National Cancer Institute 66(6): 1191–1308.
- Hanahan D and Weinberg RA (2000) The hallmarks of cancer. Cell 100: 57–70.
- Harvard Center for Cancer Prevention (1996) Human Causes of Cancer. Cambridge, MA: Harvard School of Public Health.
- Hayes RB, Songnian Y, Dosemeci M, and Linet M (2001) Benzene and lymphohematopoietic malignancies in humans. American Journal of Industrial Medicine 40: 117–126.
- Higginson J and Muir CS (1977) Determination of the importance of environmental factors in human cancer: The role of epidemiology. Bulletin du Cancer 64(3): 365–384.
- Siemiatycki J, Richardson L, Straif K, et al. (2004) Listing occupational carcinogens. Environmental Health Perspectives 112(15): 1447–1459.
- Wartenberg D, Reyner D, and Siegel Scott C (2000) Trichloroethylene and cancer: Epidemiologic evidence. Environmental Health Perspectives 108(S2): 161–176.
- Zahm SH and Ward MH (1998) Pesticides and childhood cancer. Environmental Health Perspectives, Supplement 106(3): 893–908.
- Blair A and Zahm S (1995) Agricultural exposures and cancer. Environmental Health Perspectives, Supplement 103(8): 205–208.
- Carpenter DO, Arcaro K, and Spink DC (2002) Understanding the human health effects of chemical mixtures. Environmental Health Perspectives, Supplement 110: 25–42.
- Huff J (1993) Chemicals and cancer in humans: First evidence in experimental animals. Environmental Health Perspectives 100: 201–210.
- Gouveia-Vigeant T and Tickner J (May, 2003) Toxic Chemicals and Childhood Cancer: A Review of the Evidence. Lowell, MA: Lowell Center for Sustainable Production, University of Massachusetts.
- Janssen S, Solomon G, and Schettler T (n.d) CHE Toxicant and Disease Database. https://www.healthandenvironment.org/our-work/toxicant-and-disease-database/
- Levy BS, Wagner GR, Rest KM, and Weeks JL (eds.) (2005) Preventing Occupational Disease and Injury, 2nd edn. Washington, D.C.: American Public Health Association.
- National Research Council (2005) BEIR VII-Phase 2, Health Risks from Exposure to Low Levels of Ionizing Radiation. Washington, D.C.: The National Academies Press.
- Rothman KJ (2002) Epidemiology – An Introduction. Oxford, UK: Oxford University Press.
- Schottenfeld D and Fraumeni JF (2006) Cancer Epidemiology and Prevention, 3rd edn. New York: Oxford University Press.




