View sample cancer research paper on esophageal cancer. Browse other research paper examples for more inspiration. If you need a thorough research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Introduction
Esophageal cancer is a malignant tumor that develops in the inner layers of the mucosal lining of the esophagus, the hollow, muscular tube that transports food and liquid from the pharynx to the stomach (Figure 1). The length of the esophagus in a normal adult is approximately 25 cm or about 10 inches. There are two major types of esophageal cancer, squamous cell carcinoma of the esophagus (SCCE) and adenocarcinoma of the esophagus (ACE), which share a poor prognosis but have rather distinct histopathologic and epidemiologic profiles. SCCE, which can arise in any part of the esophagus, arises from thin, flat, squamous cells that line the esophagus. ACE, usually found in the lower part of the esophagus near the stomach, forms in glandular cells (not normally present in the lining of the esophagus) that replace a section of squamous cells, a condition called Barrett’s esophagus. Although the risk of ACE in patients with Barrett’s is small (less than 1% per year), people with Barrett’s esophagus are estimated to be at a 30-fold to 125-fold higher risk of developing ACE than people who do not have this condition.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
Demographic Features
Incidence And Mortality Rates
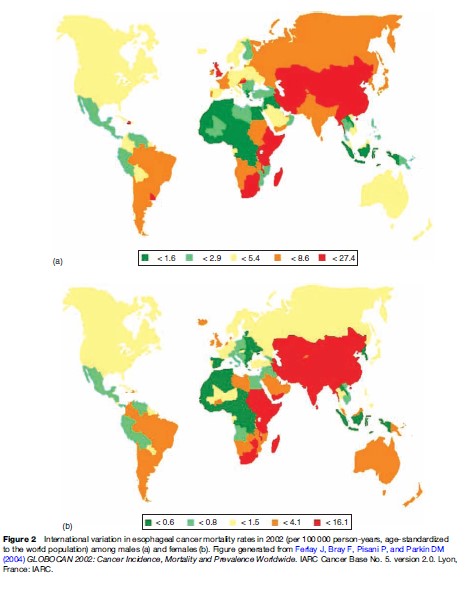
Esophageal cancer is known for its marked variation by geographic region, ethnicity, and gender. The most recent estimates of global cancer incidence indicate that esophageal cancer is the eighth most frequent cancer in the world (some 315 000 new cases a year in men and 147 000 in women), but because of its poor prognosis, it is the sixth most common cause of cancer death (261 000 deaths a year among men and 125 000 among women) (Ferlay et al., 2004). Fatality rates are so high that age standardized rates per 100 000 are generally comparable for incidence (11.5 for men, 4.7 for women) and mortality (9.9 for men, 3.9 for women). Some of the highest mortality rates in both men (Figure 2a) and women (Figure 2b) occur in the ‘Asian esophageal cancer belt,’ which stretches from northern Iran and central Asia (including Turkmenistan and Kazakhstan) into northern and western China. No population-based tumor registries operate in this region, and true incidence rates are unknown. Other areas of high mortality are found in southern and eastern Africa.
Incidence Trends And Patterns
International differences in esophageal cancer incidence rates are striking (Figure 3). Recent esophageal cancer rates varied by almost sevenfold among males, from 17.2 cases per 100 000 population per year in Calvados, France to 2.5 in Israel. Rates were also relatively high among men in Japan (14.4), Bas-Rhin, France (13.6), Scotland (11.6), and U.S. Blacks (10.7).
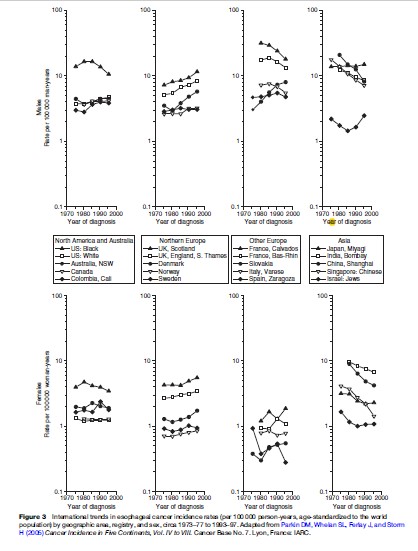
Recent rates among females varied over 20-fold, from 6.6 in India to 0.3 in Spain. Rates were also relatively high in Scotland (5.5), China (4.2), U.S. Blacks (3.5), and England (3.4). The ranking of rates among females did not mirror that among males, but rates in all areas were higher in males than in females. Male:female rate ratios varied from less than 2 in India and China to more than 6 in Japan, Italy, and Calvados, France and more than 10 in Spain, Bas-Rhin, France, and Slovakia. Rates in all populations rose consistently with age (Parkin et al., 2005).
Among men, incidence rates declined by 24–60% over most of the period 1973–1977 to 1993–1997 in U.S. Blacks, France, Italy, India, China, and Singapore; increased by 20–98% in U.S. Whites, Australia, Scotland, England, Denmark, and Norway; and more than doubled in Slovakia.
Although rates among females tended to fluctuate more over time due to the smaller number of cases, the trends were broadly similar to those in males. Incidence rates fell by 27–65% in U.S. Blacks, India, China, and Singapore, whereas they rose 21–83% in Scotland, England, Denmark, Norway, and Slovakia.
These divergent incidence trends and patterns reflect the changing frequencies of SCCE and ACE. Incidence of SCCE, which tends to be higher in developing countries and U.S. Blacks, appears to be falling, whereas the incidence of ACE, which tends to be higher in more developed countries and in U.S. Whites, has been steadily increasing. Examples of these trends are presented in Figure 4, which demonstrates the impact of changing rates of SCCE and ACE on total esophageal cancer rates among U.S. black and white men over the time period 1974–78 to 1999–2003.
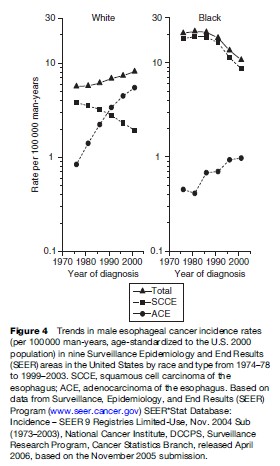
Rates per 100 000 among U.S. Blacks peaked at 21.6 in 1979–83 and then began a marked decline, reaching 10.7 in 1999–2003. Rates per 100 000 among white men, however, increased steadily from 5.7 in 1974–78 to 8.2 in 1999–2003. The dramatic decrease in total esophageal cancer rates for black men was driven by the 54.7% drop in the incidence of SCCE (from 19.0 in 1979–83 to 8.6 in 1999–2003), which is more than eight times as common as ACE in Blacks. The incidence of SCCE also decreased 50% among white males (from 3.8 in 1974–78 to 1.9 in 1999–2003). In contrast, among white males, the incidence of ACE rose almost seven-fold from 0.8 in 1974–78 to 5.5 in 1999–2003. With the decrease in SCCE and the increase in ACE, the rates for ACE among white men surpassed those for SCCE during the period 1989–93. Rates of ACE among black men more than doubled over the past three decades; however, the rates of SCCE remain considerably higher.
Etiologic Factors
The established or suspected risk or protective factors for SCCE and ACE are listed in Table 1 and explained in greater detail below.
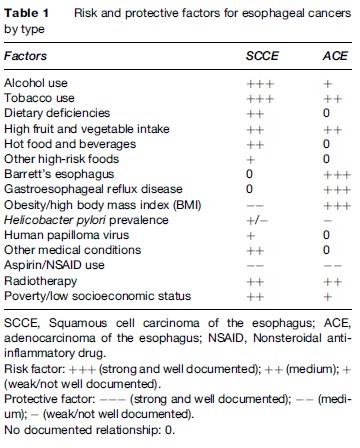
Alcohol Use
There is strong epidemiologic evidence that excessive consumption of alcoholic beverages is a major cause of SCCE, especially in Western populations. In some developing countries with exceptionally high rates of SCCE, including parts of Africa, Iran, and China, alcohol drinking historically had not been shown to be a risk factor. However, alcohol has recently been identified as a risk factor in southern India, Hong Kong, and Shanghai, China. Although only a few studies have investigated esophageal cancer risk associated with alcohol use among nonsmokers, the dose–response gradient for alcohol consumption remains strong when analyses are restricted to lifelong nonsmokers. Variability in risk by type of alcoholic beverage is thought to reflect drinking habits that are culturally or economically determined. Generally, the beverage most frequently consumed by the population is the one most strongly associated with the risk of SCCE. The formerly widespread habit of drinking hot alcoholic beverages, particularly hot calvados, in France, probably contributed both to the higher rates of SCCE there than among men in other European countries and to the recent downward trends in incidence depicted in Figure 3. Although alcohol is strongly related to the risk of SCCE, the mechanisms responsible for its carcinogenicity have not been identified but may include acting as a local chronic irritant, promoting dietary deficiencies, and enhancing susceptibility to the effects of tobacco or other carcinogens, or the role of acetaldehyde (a metabolite of alcohol and a recognized animal carcinogen).
Alcoholic beverage consumption appears to play at most a minor role in the etiology of ACE.
Tobacco Use
Tobacco use, regardless of form, is a major risk factor for SCCE in most parts of the world. Significant positive trends in risk have been associated with both the intensity and the duration of cigarette smoking. Reductions in risk in the neighborhood of 50% have been seen for exsmokers compared to current smokers, along with an inverse effect with time since cessation of smoking. In addition to traditional cigarettes, cigars, pipes, snuff, and chewing tobacco, elevated risks have also been found in India associated with smoking bidi (native cigarettes of coarse tobacco in a dry termburni leaf) and the chewing of pan (a mixture of betel leaf, sliced areca nut, and aqueous shell lime) and pan-tobacco. In South America and Europe, lower risks of SCCE have been reported among smokers of blond rather than black tobacco; the smoke of black tobacco contains higher levels of carcinogens and mutagens. A number of known or suspected carcinogens such as nitrosamines, benzo[a]pyrene, and benzene have been identified in tobacco smoke, but the specific agents responsible for SCCE and their mechanisms of action are not clear. It is likely that declines in the prevalence of smoking since the 1960s, especially among men in the United States, may have contributed to the downward trends of SCCE (Brown and Devesa, 2002).
In Western Europe and North America, 80 to 90% of the risk of SCCE has been attributed to use of alcohol and tobacco. Alcohol and tobacco appear to act independently, with the importance of each factor depending on characteristics of the population. Alcohol seems to be more important in a population of heavy drinkers, whereas tobacco appears to be more important in a population of heavy smokers. In most studies, the highest risk of SCCE is seen in heavy consumers of both alcohol and tobacco, often consistent with a multiplicative interaction. For a public health standpoint, it is clear that reduction in alcohol and tobacco use would markedly lower the incidence of SCCE (Brown et al., 2001).
Although smoking is a less potent cause of ACE than SCCE, elevated risks of ACE have been documented, especially among heavy smokers. Contrary to the findings for SCCE, smoking cessation does not appear to diminish risk; ACE risk remains elevated 30 or more years after stopping smoking.
Dietary Factors
Deficiencies
The variations in SCCE incidence around the world have been linked to dietary insufficiencies. Historically, SCCE has been linked to Plummer-Vinson syndrome, which is associated with vitamin and iron deficiencies, and to celiac disease, a malabsorption syndrome often characterized by malnutrition. However, in some populations it has been difficult to disentangle the influence of dietary factors from the more potent effects of alcohol and tobacco. Consumption of alcoholic beverages and tobacco products can interfere with the intake and metabolism of a variety of nutrients and can also be sources of N-nitroso compounds that may increase the risk of SCCE.
Fruit And Vegetable Intake
The most consistent dietary finding for SCCE has been the protective effect of consumption of fruits and vegetables, especially those eaten raw, which contain a variety of micronutrients and dietary components. Studies have suggested a protective effect of vitamin C, which blocks the endogenous formation of N-nitroso compounds. Other nutrients which may be protective include alpha and beta-carotene, lycopene, folate, vitamin E, vitamin B6, vitamin B12, zinc, selenium, and flavonoids.
Various foods, food groups, and nutrients have also been related to the risk of ACE, but the most consistent finding is a protective effect of fruits, vegetables, and fiber. Nutrients that may be protective against ACE include vitamin C, vitamin E, beta-carotene, vitamin B6, vitamin B12, and folate.
Other Foods And Beverages
A number of studies have suggested that excess risks of SCCE may be associated with consumption of burning hot soup, gruel, porridge, tea (especially mate), and other beverages in various populations around the world.
Suspected risk factors for SCCE include foods that are sources of heterocyclic amines formed during cooking (e.g., barbecued or fried meats, especially red meat) or N-nitroso compounds or their precursors nitrates and amines (e.g., cured or processed meat, moldy breads and cereals, pickled vegetables, and salted fish).
Medical Conditions And Treatments
Gastro-Esophageal Reflux Disease
The reflux of gastric acid into the esophagus, which commonly presents as chronic heartburn, in gastroesophageal reflux disease (GERD) may stimulate the esophageal mucosa to develop metaplastic columnar epithelium, which is characteristic of the ACE precursor lesion, Barrett’s esophagus, found in almost all patients with ACE. No clear association has been found between drugs used to treat GERD and the risk of ACE (Blot et al., 2006).
Obesity
Populations at high risk for SCCE tend to be poorly nourished. Within a population, risk tends to increase as body mass index (BMI) (weight in kg/height in meters2) decreases.
In contrast, risk of ACE tends to increase as BMI rises, with clinically obese individuals (BMI > 30) having a substantially higher risk of ACE than individuals of normal or lower weight (BMI < 25). Data emerging from recent studies suggest that the relationship with obesity may account for part of the upward trend in ACE incidence (Kubo and Corley, 2006). Although the exact mechanism is unclear, it may be related to the higher prevalence of GERD in obese individuals.
Helicobacter pylori
Infection with H. pylori, a risk factor for stomach cancer, appears to be associated with a reduced risk of ACE. Whether H. pylori infection is related to risk of or protection from SCCE is unclear. Further investigations are needed to determine whether the decreasing prevalence of H. pylori infection may be related to the upward trend for ACE (Blot et al., 2006).
Other Medical Conditions And Treatments
The possible role of human papilloma virus (HPV) infection in the etiology of SCCE has been controversial. It appears to be related to SCCE risk in areas with exceptionally high incidence of esophageal cancer such as China, Iran, South Africa, and South American, but not in the United Kingdom or North America. Increased SCCE risks have been reported with chemical injuries to the esophagus and with medical conditions such as pernicious anemia, achalasia, some autoimmune diseases, and following gastrectomy. Regular use of aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs) appears to be related to a reduced risk of both SCCE and ACE. Exposure to ionizing radiation, particularly among women who received radiotherapy for breast cancer, has been linked to excess risks of both SCCE and ACE.
Poverty/Low Socioeconomic Status (Ses)
The highest rates of SCCE are generally found in impoverished areas of the world. Within a population, the risk of SCCE is greatest among those with the lowest SES. Low SES, whether measured by education, income, or occupation, is a surrogate for a set of complex lifestyle and environmental factors (e.g., poor living conditions, exposure to infectious or chemical agents, workplace hazards, poor nutrition, cigarette smoking, excessive alcohol consumption, and limited access to medical care).
Low SES has also been related to excess risk of ACE, but the effect is less pronounced than for SCCE.
Diagnosis
Signs And Symptoms
Symptoms of esophageal cancer generally do not appear until the tumor has grown large enough to obstruct part of the esophagus. The most common sign of esophageal cancer is dysphagia or difficulty swallowing solids or liquids. As the tumor grows and becomes locally invasive, additional symptoms may include:
- Odynophagia (painful swallowing)
- Regurgitation of food
- Indigestion and heartburn
- Weight loss
- Chest pain behind the sternum (unrelated to eating)
- Hoarseness
- Cough
- Vomiting blood.
Diagnosis And Staging Tests
Tests to rule out esophageal cancer include:
- A barium-swallow examination (also called an upper GI series or esophagogram): the patient drinks a barium solution, which coats the surface of the esophagus so that any irregularities on the lining of the esophagus show up when a series of X-rays are taken.
Upper endoscopy (also called esophagoscopy): a thin, flexible, lighted tube (called an endoscope or esophagoscope) with a camera on the end is passed through the mouth or nose and down the esophagus to look for abnormal areas. If an abnormal area is found, a biopsy specimen will be taken and examined for signs of cancer.
Once a diagnosis of esophageal cancer has been confirmed, the doctor will need to perform staging tests, important in determining treatment, to ascertain whether cancer cells have spread within the esophagus or metastasized outside the esophagus to nearby organs and lymph nodes. Common staging tests and procedures may include:
- Computed tomography (CT) or computerized axial tomography (CAT): a series of detailed X-ray pictures are taken at different angles inside the body and integrated by a computer to produce three-dimensional images.
- Ultrasonography (also called ultrasound): high-energy sound waves or ultrasound are bounced off internal tissues (including lymph nodes) or organs, forming a picture called a sonogram.
- Endoscopic ultrasound (EUS) (also called endosonography): an endoscope with a small ultrasound probe on the tip that bounces sound waves off the walls of the esophagus is passed through the mouth and into the esophagus. This procedure is very good at determining the depth of the tumor in the esophageal wall and whether adjacent lymph nodes are involved.
- Positron emission tomography (PET) scan: a small amount of radioactive glucose is injected into a vein. The scanner makes detailed pictures of where the glucose is used in the body. Since the glucose is taken up more rapidly by cancer cells than normal cells in the body, it can be used to pinpoint cancer cells.
- Magnetic resonance imaging (MRI): intense magnetic fields and radio-frequency waves are used to provide real-time, three-dimensional views of the human body.
- Other procedures sometimes used for staging include thoracoscopy, laparoscopy, bronchoscopy, and laryngoscopy.
Treatment
Treatment for esophageal cancer is determined by:
- The stage of the cancer, which ranges from Stage 0 (carcinoma in situ), in which cancer is confined to the innermost lining of the esophagus, to Stage IV (metastatic cancer), in which the cancer has spread to distant organs.
- The size of the tumor.
- The general health of the patient.
Cure is possible if the cancer is diagnosed at an early stage and the patient is healthy enough to undergo surgery. Unfortunately, symptoms usually do not occur until the tumor is large; therefore, most patients present with locally advanced or metastatic disease. For these patients, cure is unlikely; the goal of treatment is generally to relieve or palliate symptoms and prolong survival. The treatments used to treat early esophageal cancer are surgery, radiation, and chemotherapy or some combination thereof. These treatments can also be used to relieve symptoms and reduce pain.
Surgery
The most common treatment for esophageal cancer is the surgical removal or resection of the cancerous part of the esophagus and the nearby lymph nodes, a procedure called esophagectomy. There is a significant mortality following major esophageal resections unless done in a specialized center.
Chemotherapy
Chemotherapy involves the oral or intravenous use of drugs to kill or inhibit the growth of esophageal cancer. Chemotherapy by itself will not cure esophageal cancer, but it is often used in combination with surgery and/or radiation. It can be given pre- and post-operatively.
Radiation Therapy
External radiation therapy is commonly used to treat esophageal cancer by using a beam of high-energy rays to shrink the tumor and kill the cancer cells. Radiation therapy alone will not cure esophageal cancer. It is often combined with surgery and/or chemotherapy.
Survival
Five-year relative survival is an estimate of the chance of surviving the effects of cancer five years after diagnosis. It is calculated by adjusting the survival rate to remove all causes of death except cancer. Although survival among patients diagnosed with esophageal cancer is poor, significant improvements in five-year relative survival in the United States and Europe have occurred over the past three decades (Table 2). Five-year relative survival for those diagnosed during 1995–99 was substantially higher for U.S. Whites than for U.S. Blacks (15.3% vs. 9.3%). Survival is similar for patients with SCCE and ACE. There is a strong decreasing gradient in survival with increasing extent of disease at diagnosis for all esophageal cancer patients. Recent five-year survival rates for U.S. patients diagnosed with esophageal cancer ranged from 34% for those with localized disease to 17% for regional disease to 3% for distant disease (Ries et al., 2006).
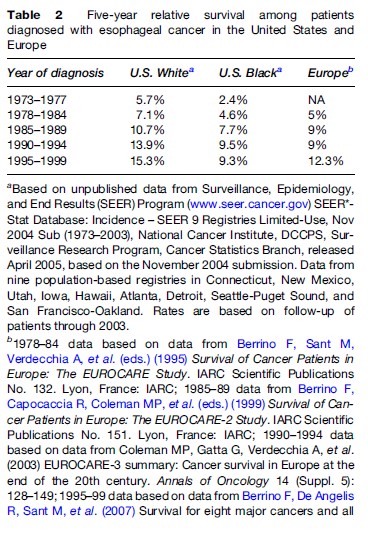
Pooled estimates of five-year relative survival from up to 23 European countries have generally been similar to the five-year relative survival among U.S. Blacks, but the most recent results for Europe suggest a value intermediate between U.S. Blacks and Whites (Table 2). There appears to be substantial variation in the five-year relative survival among residents of developing countries when compared to U.S. rates during the same time periods (Sankaranarayanan et al., 1998). For example, survival rates of 4% among residents of Quidong, China and Chiang Mai, Thailand diagnosed during 1982–91 and 1983–92, respectively, were lower than comparable rates among U.S. Blacks; survival rates of 7% among residents of Madras, India diagnosed during 1984–89 were similar to survival rates among U.S. Blacks; and survival rates of 11% among residents of Shanghai, China diagnosed during 1988–91 resembled survival rates in U.S. Whites.
Public Health Significance And Global Importance
Esophageal cancer is the eighth most common cancer worldwide and the sixth most common cause of cancer death. It is a highly fatal tumor if not detected early and treated aggressively with surgery, either alone or in combination with radiotherapy and/or chemotherapy. There is no cost-effective method for screening asymptomatic patients; however, it is recommended that patients with Barrett’s esophagus, the precursor condition for adenocarcinoma of the esophagus, undergo surveillance endoscopy and biopsy. Decreasing use of alcohol, tobacco, and very hot foods and beverages, and increasing intake of fresh fruits and vegetables may lead to lower incidence of squamous cell carcinoma of the esophagus in some populations. Reasons for the rapid rise in adenocarcinoma of the esophagus, especially in several Western populations, remain elusive but may be related to epidemic increases in the proportion of obese adults and the subsequent increases in gastro-esophageal reflux disease. Treatment is generally palliative rather than curative, because most esophageal cancers are not detected until the tumor has grown large enough to inhibit swallowing, at which point it is not usually susceptible to radical surgery of curative intent. Reduction in the burden of esophageal cancer will only come from primary prevention by reducing exposure to known risk factors, especially tobacco and alcohol, and increasing exposure to protective factors, especially dietary fruit, vegetables, and fiber. This is because screening offers no prospect of reducing mortality except in the very small subpopulation with the premalignant condition Barrett’s esophagus, and the natural history of the disease precludes early clinical diagnosis in the vast majority of cases.
Bibliography:
- Berrino F, Sant M, Verdecchia A, et al. (eds.) (1995) Survival of Cancer Patients in Europe: the EUROCARE study. IARC Scientific Publications No. 132. Lyon, France: IARC.
- Berrino F, Capocaccia R, Coleman MP, et al. (eds.) (1999) Survival of Cancer Patients in Europe: the EUROCARE-2 study. IARC Scientific Publications No. 151. Lyon, France: IARC.
- Berrino F, DeAngelis R, Sant M, et al. (2007) Survival for eight major cancers and all cancers combined for European adults diagnosed in 1995–99: results of the EUROCARE-4 study. Lancet Oncology 8: 773–783.
- Blot WJ, McLaughlin JK, and Fraumeni JF Jr. (2006) Esophageal cancer. In: Schottenfeld D and Fraumeni JF Jr. (eds.) Cancer Epidemiology and Prevention, 3rd ed., pp. 697–706. New York: Oxford University Press.
- Brown LM, Hoover R, Silverman D, et al. (2001) Excess incidence of squamous cell esophageal cancer among US black men: role of social class and other risk factors. American Journal of Epidemiology 153: 114–122.
- Brown LM and Devesa SS (2002) Epidemiologic trends in esophageal and gastric cancer in the United States. Surgical Oncology Clinics of North America 11: 235–256.
- Coleman MP, Gatta G, Verdecchia A, et al. (2003) EUROCARE-3 summary: cancer survival in Europe at the end of the 20th century. Annals of Oncology 14(Supplement 5): 128–149.
- Ferlay J, Bray F, Pisani P, and Parkin DM (2004) GLOBOCAN 2002: Cancer Incidence, Mortality and Prevalence Worldwide. IARC Cancer Base No. 5. version 2.0. Lyon, France: IARC.
- Kubo A and Corley DA (2006) Body mass index and adenocarcinomas of the esophagus or gastric cardia: a systematic review and meta-analysis. Cancer Epidemiology Biomarkers and Prevention 15: 872–878.
- Parkin DM, Whelan SL, Ferlay J, and Storm H (2005) Cancer Incidence in Five Continents Vol. IV to VIII. Cancer Base No. 7. Lyon, France: IARC.
- Ries LAG, Harkins D, Krapcho M, et al. (eds.) (2006) SEER Cancer Statistics Review, 1975–2003. Bethesda, MD: National Cancer Institute.
- Sankaranarayanan R, Black RJ, and Parkin DM (1998) Cancer Survival in Developing Countries. IARC Scientific Publications No. 145. Lyon, France: IARC.
- Surveillance, Epidemiology, and End Results (SEER) Program (2006) SEER*Stat Database: Incidence – SEER 9 Registries Limited-Use, Nov 2005 Sub (1973–2003), National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch, released April 2006, based on the November 2005 submission.
- Ajani JA, Curley SA, Janjan NA,, and Lynch PM (eds.) (2005) Gastrointestinal Cancer. M.D. Anderson Cancer Care Series. New York: Springer.
- DHHS/PHS/NIH/NCI (2000) What You Need to Know about Cancer of the Esophagus. NIH Publication No. 00–1557. Bethesda, MD: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Cancer Institute.
- Enzinger PC and Mayer RJ (2003) Esophageal cancer. New England Journal of Medicine 349: 2241–2252.
- Posner MC (ed.) (2002) Surgical Oncology Clinics of North America Contemporary Management of Esophageal and Gastric Carcinoma 11(2).
- Posner MC, Vokes EE,, and Weichselbaum RR (eds.) (2002) American Cancer Society Atlas of Clinical Oncology. Cancer of the Upper Gastrointestinal Tract. Hamilton, Ontario: BC Decker.
- Posner MC, Forastiere AA, and Minsky BC (2005) Cancers of the gastrointestinal tract: Cancer of the esophagus. In: DeVita VT,
- Hellman S,, and Rosenberg SA (eds.) Cancer: Principles and Practice of Oncology, 7th edn. Philadelphia, PA: Lippincott Williams & Wilkins.
- https://www.cancer.org/ – American Cancer Society.
- https://www-dep.iarc.fr/ – International Agency for Research on Cancer (IARC).
- https://www.cancer.gov/ – National Cancer Institute, U.S. National Institutes of Health.
- https://seer.cancer.gov/ – SEER Surveillance Epidemiology and End Results.




