View sample cancer research paper on cancer mortality. Browse other research paper examples for more inspiration. If you need a thorough research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Introduction
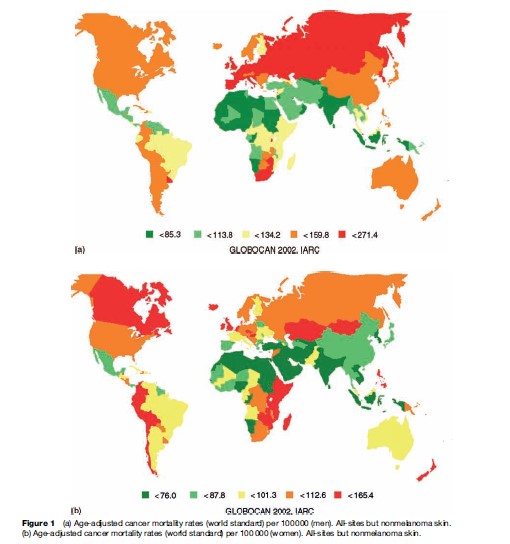
According to all-cause estimates from the World Health Organization, 7.6 million people died from cancer in the year 2005, representing approximately 13% of deaths from all causes worldwide (WHO, 2006). The proportion of all annual deaths attributed to cancer varies considerably between and within the world regions, from around 23% in North America to only 4% in Africa (Ferlay et al., 2004). Age-adjusted (world standard) mortality rates of cancer in men of around 138 per 100 000 are about 50% higher than rates observed for women. Generally, rates are also higher in more developed areas: 169.8 and 119.3 per 100 000 for men in more and less developed countries, respectively, compared with 102.5 and 83.1 per 100 000 for women, respectively. The global map of overall cancer mortality by sex in Figure 1(a), (b) illustrates the variability between and within world areas, with high rates observed both among men and women in certain countries in each of the world’s continents.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
Overall, the most common cause of death from cancer is lung cancer, which accounts for close to two-fifths of all deaths due to cancer. Stomach cancer is in second place (10%), followed by liver cancer (9%). There are some differences in the five leading causes of cancer mortality between sexes: In men, deaths from cancers of the lung, stomach, liver, colorectal, and esophagus are the most frequent, while in women, breast, lung, cervix, stomach, and colorectal cancer dominate. Except for esophageal cancer (in men), these cancer sites also represent the cancers with the highest incidence globally (Ferlay et al., 2004), although the specific incidence ranking is somewhat different given the variation in the survival rates from these tumors.
In this research paper we discuss the properties of cancer mortality, the primary data sources, and the availability and quality of data between countries and over time, and the ensuing difficulties with interpretation. We also provide some selected descriptions of cancer mortality patterns by sex and populations. In particular, we focus on lung, stomach, and liver cancer, the three major causes of death from cancer at the global level. This research paper concludes with an assessment of the application of mortality trends as an indicator of the progress against cancer.
Characteristics Of Cancer Mortality
Definition
Cancer mortality is expressed either as number of cancer deaths occurring, or as a mortality rate, i.e., the number of cancer deaths per 100 000 persons per year. Cancer mortality provides a measure of the impact of cancer in a given population. Mortality data derive from vital registration systems, where usually a medical practitioner certifies the fact and cause of death. The International Classification of Diseases (ICD) provides a uniform system of nomenclature and coding and a recommended format for the death certificate.
Cancer mortality is the product of the incidence and the case fatality rate of a given cancer. Death rates estimate the average risk to the population of dying from a specific cancer, whereas the case fatality rate, the inverse of survival, represents the proportion of individuals with specific cancer that will die from it within a specified time. Thus, for stable trends over time, a case fatality rate of 0.25 (equal to a survival of 0.75) would yield a mortality rate one-quarter that of incidence.
Relation To Incidence And Survival
Cancer incidence data tend to be available in fewer populations and for shorter time periods than cancer mortality data. Mortality data are therefore sometimes used as a practical surrogate for incidence. Still, mortality data do not always reflect incidence given its dependence on both incidence and fatality. For certain cancers associated with reasonable survival, prognosis over several decades may have improved. The concomitant declines in case fatality may then result in substantial and misleading variability in the temporal trends of the incidence and mortality rates. Mortality is better suited as a proxy for incidence of cancers with poor survival and for which there is a short interval between diagnosis and death (e.g., lung cancer): Here the variability between the two indicators will be much less heterogeneous.
Historically there have also been some concerns with incidence. In their landmark study of environmental causes of cancer in the United States, published in 1981, Doll and Peto utilized mortality rather than incidence, citing that the latter was a more complex statistic to interpret. They were concerned about two factors. First, and in addition to the artifacts associated with changing efficiency of the registration process, they considered the effects of changing practice in classification of cancers associated with different rates of fatality and the spread of screening tools that detect cases earlier as having had a large impact on the record incidence of many cancer types. Second, they noted that incidence, while of great value, did not remove the need for mortality data, necessary – in combination with incidence – to detect improvements in therapy, and partly to verify the validity of incidence trends. While Doll and Peto’s circumvention of the available incidence data drew some criticism (Clemmesen and Nielsen, 1982; Devesa et al., 1984), there has long been consensus that combined description of trends in incidence, mortality, and survival often serves to confirm and clarify understanding of the underlying biological and epidemiological processes, as well as restate the relative strengths and weaknesses of each measure in interpretation (Boyle, 1989). However, mortality rates are effective indicators of disease outcome, for instance in determining the beneficial effects of a specific treatment regimen at the population level.
Sources Of Data, Availability, And Estimation
Mortality data are commonly available from national statistics offices by year of death, 5-year age group, and sex. Their relative advantage over incidence stems from their more comprehensive availability. Partly for this reason, temporal analyses of cancer mortality tend to be more common than those of incidence in the medical and epidemiological literature. The national data are combined in the WHO mortality databank, which contains data series going back in time for 70 countries. Country-specific estimates worldwide are available (presently for 2002) from the GLOBOCAN database.
The WHO Mortality Databank
The WHO mortality databank is an updated database of cause-specific national mortality for many countries in both developed and developing areas, and contains data as far back as the 1950s. The mortality data were previously published in the World Health Statistics Annual, but are now available on WHO’s website. Online tables and figures are available from the International Agency for Research on Cancer (IARC) website containing data from 1955 to 2004. The WHO data are based on deaths registered in national vital registration systems, with underlying causes of death coded using ICD9 and ICD10. The number of registered deaths are coded by cause, sex, and age for each country and year. Some discussion of the availability and quality of death registration data, assessed by timeliness, completeness and coverage of registration, and proportion of deaths assigned to ill-defined causes for national mortality data in general, is available from the Bulletin of the World Health Organization (Mathers et al., 2005).
GLOBOCAN 2002
The IARC compiles the GLOBOCAN database, a database that in addition to cancer mortality data also provides data on cancer incidence and prevalence (Ferlay et al., 2004). The data are presented by sex for 175 countries worldwide for 27 cancer sites. GLOBOCAN currently presents cancer mortality estimates by world country for the year 2002, i.e., GLOBOCAN 2002.
Several data sources and methods have been utilized to provide these estimates depending on the availability of data, the level of detail, and its accuracy. National mortality data have been applied whenever available. If the data were known to underestimate the number of deaths, this has been corrected for in the published data. If no national mortality data were available, local or regional data were used for the entire country. Finally, for countries with no mortality data or data considered of poor quality, the estimates are based on incidence and country or region-specific survival. Where cancer data were neither available nationally nor regionally, mortality was estimated from the average of selected neighboring countries within the same regions.
Since cancer data are always collected and compiled sometime after the events to which they relate, the most recent statistics are only available several years after the end of the corresponding year of death. The cancer deaths estimated in GLOBOCAN 2002 are generally based on the application of population data derived from 1997 to 2000. As the sources of data are continuously improving in quality and extent, and the methods re-evaluated, one should be careful when comparing GLOBOCAN 2002 estimates with worldwide estimates published in prior years. The estimates are not strictly comparable, and observed differences may in part be a result of changes in methodology.
Data Quality
Mortality statistics are produced according to the underlying cause of death, although this may not equate with the presence of a particular tumor. Comprehensive mortality statistics require that diagnostic data are available on decedents, which are transferred in a logical, standardized fashion to death certificates, which are then accurately and consistently coded, compiled, and analyzed. Death registrations require that the correct diagnosis is written on the death certificate and further that this diagnosis is then certified as the underlying cause of death.
Many studies have investigated the accuracy of death certificate diagnoses in vital statistics data, comparing cause of death entered on the death certificate with a reference diagnosis derived from autopsy reports, detailed clinical records, or cancer registry data. Such studies have shown that the level of accuracy of the stated cause of death declined as the precision in the diagnosis increased; although the total number of deaths from cancer of all types may only be slightly underestimated, the corresponding distribution by site of cancer may be associated with more substantial errors. A tendency to overregister nonspecific diagnoses instead of the correct location (e.g., large intestine instead of rectum) has been noted, and the accuracy tends to be lower in those dying at older ages.
Illustrative examples include a study by Grulich and co-workers (1995) on cancer trends in England and Wales 1970–90 in the 75–84 year age group. They found that the rise in all-cancer mortality in the elderly was in part due to increasing lung cancer mortality, but data artifacts were responsible for much of the increase in the other commonly specified cancers (Grulich et al., 1995). Another well-known study by Percy and co-workers (1981) compared death certificates mentioning cancer as the underlying cause of death for almost 50 000 incidence cases, contrasting detection rates (proportion of death certificates conveying the same diagnosis as that made in life) and confirmation rates (proportion of cancer deaths for which the underlying cause was confirmed by hospital diagnosis) (Percy et al., 1981). Overreporting on death certificates was indicated when the detection rate was high relative to the confirmation rate, as was the case for neoplasms of the larynx and colon, and unspecified cancers of the uterus. Conversely, underreporting was observed (confirmation high relative to detection rates) for cancers of the cervix and corpus uteri, and rectal cancers. For melanoma and breast cancer, death certification was deemed more or less correct.
There may also be quite marked differences between countries in the allocation of ICD codes to death certificate diagnoses (Percy and Dolman, 1978). As well as erroneous death certification, mortality data are affected by changes in coding practice.
Worldwide Cancer Mortality: Variations By Cancer Site And Sex
The estimated eight million people dying from cancer in 2005 translates to approximately one in eight deaths worldwide, irrespective of cause. Overall, the age-adjusted mortality rate of cancer (world standard) is higher in men: 137.5 per 100 000 compared to 92.1 per 100 000 for women. Rates vary greatly between different populations of the world as shown in Figure 1 and 2. Male all-cancer mortality rates among Western African men are almost three times lower than those for men in Central and Eastern Europe (73.5 and 197.2 per 100 000, respectively) (GLOBOCAN, 2002).
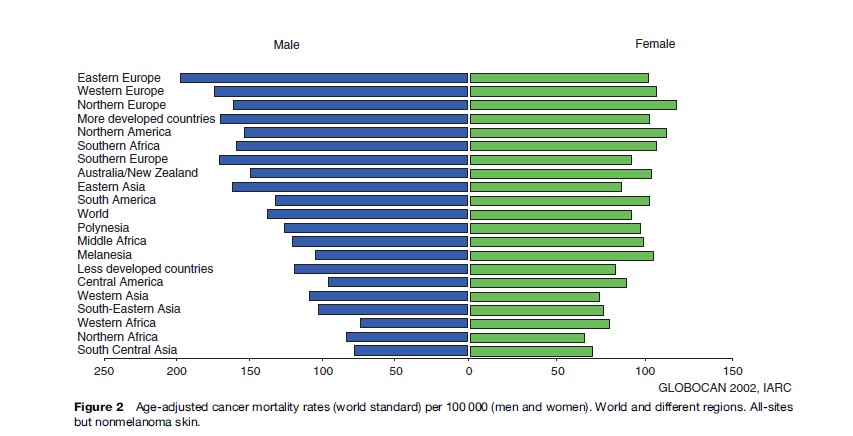
With almost 19 000 cancer deaths in 2002, rates in men in Hungary are the highest in the world: The cumulative mortality of 13.3 indicates that close to one in seven Hungarian men are dying of lung cancer before the age of 65. While the high mortality rates in Hungary and other Eastern Europe countries (Slovakia, Czech Republic) largely reflect smoking patterns and lung cancer, the patterns among women are indicated by a lesser geographical clustering of high rates and a lesser degree of clear cancerspecific causes: All-cancer mortality rates are highest for instance in Zimbabwe (165.4 per 100 000), where there are particularly high rates of cervical cancer and Kaposi sarcoma. The second-highest rates among women are observed in Denmark (148.1 per 100 000), a country with notably high mortality rates of breast and lung cancer. While mortality rates in the more developed regions of the world are three to four times as high as those of the less-developed countries, in terms of absolute numbers more than 70% of all cancer deaths in 2005 occurred in low and middle-income countries (WHO, 2006).
Compared to estimates that were given for 1990, there has been a worldwide increase in mortality rate of approximately 10% (Pisani et al., 1999). Given the current trends and an aging population, the death toll from cancer globally will likely continue to rise.
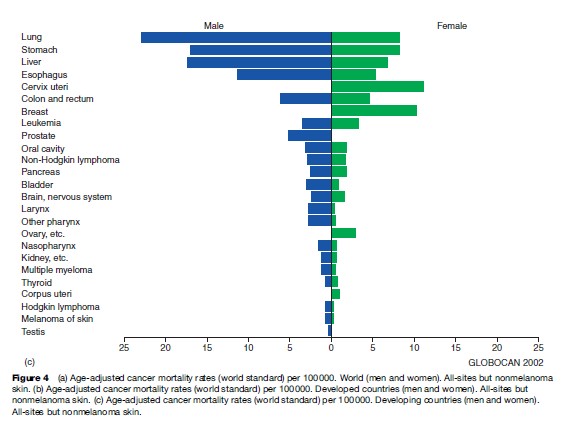
Looking at men and women together, lung cancer is the most frequent cause of death from cancer worldwide, as it is when we consider global cancer incidence. Stomach and liver cancer rank second and third, respectively. In developing countries, these cancers predominate, although cervical cancer and esophageal cancer are also important causes of cancer death. Figures 3(a), (b) present global numbers of cancer deaths for the most common cancer sites for men and women, respectively, as well as age-adjusted mortality rates worldwide. They are partitioned into developed and developing regions in Figures 4(a), (b), (c).
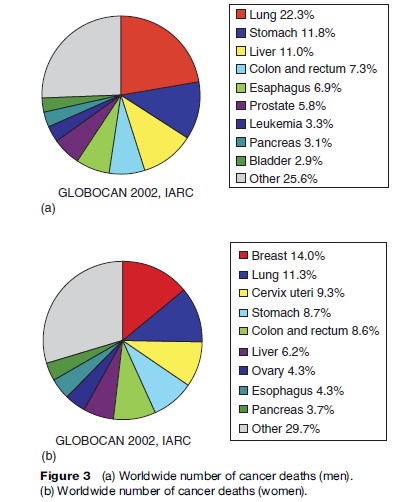
For men, lung cancer, stomach cancer, and liver cancer are the most common causes of death from cancer, followed by colorectal and esophageal cancer. For most cancer forms, mortality rates are higher for men than for women, and for all cancers combined, worldwide rates in men are about 25% higher (Figure 4(a)).
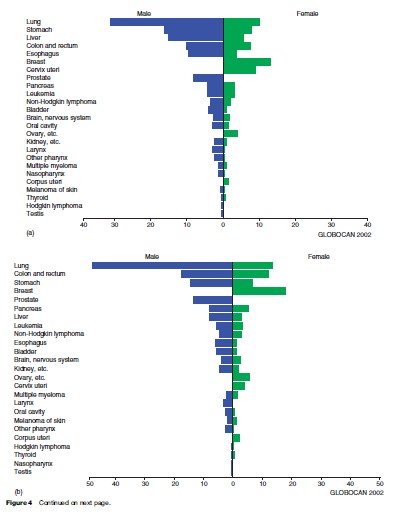
For women, breast cancer is the main cause of death from cancer, accounting for 14% of all female cancer deaths worldwide (Figure 3(b)). More than 400 000 women die of breast cancer every year; some 220 000 living in the less developed parts of the world. The age-adjusted death rate from breast cancer ranges from 29.8 and 27.8 per 100 000 in Malta and Belgium, respectively, to 2.0 per 100 000 in Haiti and 2.8 per 100 000 in Mozambique. The second to fifth most common causes of death from cancer in women worldwide are neoplasms of the lung, cervix uteri, stomach, and colorectum.
Lung Cancer
Lung cancer is the most common cause of death from cancer worldwide, with approximately 1.1 million people dying of it every year. Approximately half of the lung cancer deaths occur in less developed areas of the world. The highest age-adjusted mortality rates for males are found in Hungary (83.9), Belgium (69.9), and Poland (68.4), whereas in women the highest figures are found in Denmark (27.8 per 100 000), Northern America (26.7), and Iceland (25.2). As can be seen in Figure 5, the rates are considerably higher in developed than in developing countries.
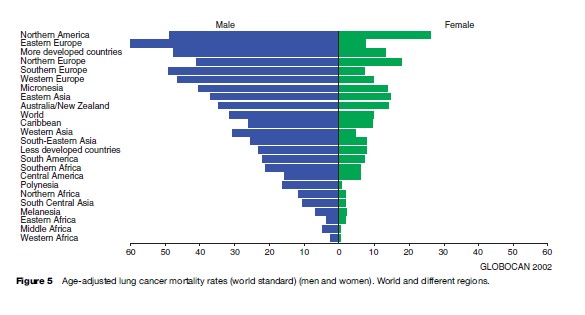
Overall, the age-adjusted mortality rate for lung cancer is more than three times higher in men than in women (31.2 and 10.3 per 100 000, respectively). Maps describing the geographical variation in lung cancer mortality rates for men and women are given in Figures 6(a), (b), respectively.
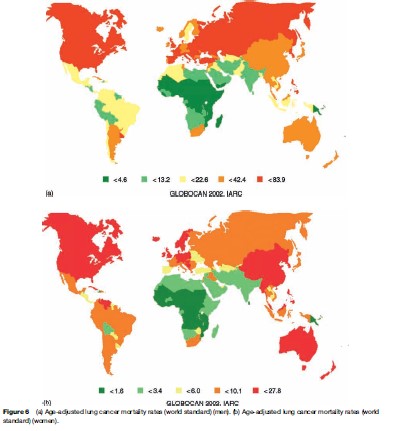
Survival following a lung cancer diagnosis is poor. The geographical and temporal variations in mortality rates therefore tend to be a good indicator of overall incidence, largely reflecting population-level changes in smoking patterns, with mortality rates by age related primarily to year of birth. Smoking prevalence has either leveled off or declined in recent decades in some developed countries, particularly among men (e.g., United States, United Kingdom), where birth cohort-specific mortality is related to the smoking habits of the same generation; and those countries where smoking was first established were also the first to observe a decline in smoking prevalence among males, followed, in the same generations of men, by a decline in risk. In most other countries, there is a continuing rise in rates, and this is most dramatic in the countries of Eastern Europe. In women, the tobacco habit has generally been adopted quite recently, or not at all. Thus, the most common picture in Western populations is of rising rates, while in many developing countries (where female smoking generally remains rare), lung cancer rates remain very low.
However, tobacco consumption is rising in the populations within the developing world (Mackay and Eriksen, 2002), and a marked increase in lung cancer mortality can therefore be anticipated in many of the highly populated regions of Africa and Asia (Peto et al., 1996).
Stomach Cancer
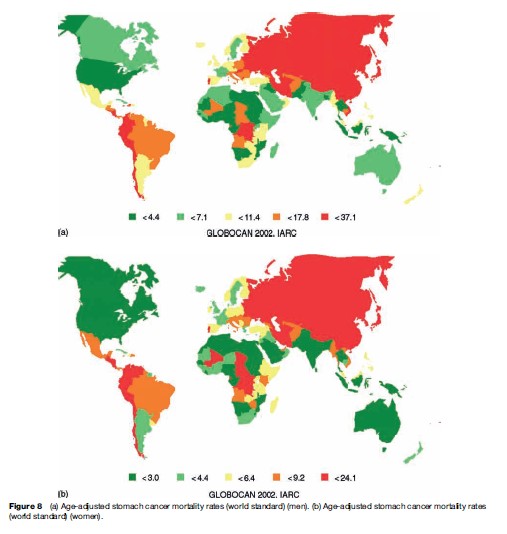
Stomach cancer is the second most common cause of death from cancer. GLOBOCAN 2002 estimates the number of stomach cancer deaths to be around 83 500 and 128 500 for women in developed and less developed countries, respectively. There were close to 170 000 deaths caused by stomach cancer in men in developed countries, compared to more than 315 500 in men in less developed areas. The global age-adjusted mortality rate for stomach cancer is 16.3 per 100 000 for men and 7.9 per 100 000 for women. As can be seen in Figures 7 and 8(a) and (b), the age-adjusted mortality rate varies greatly across different regions of the world. The large variation in stomach cancer mortality rates worldwide may be exemplified by the figure of 32.5 per 100 000 for Eastern Asian men compared to 3.2 per 100 000 for men in Western Africa.
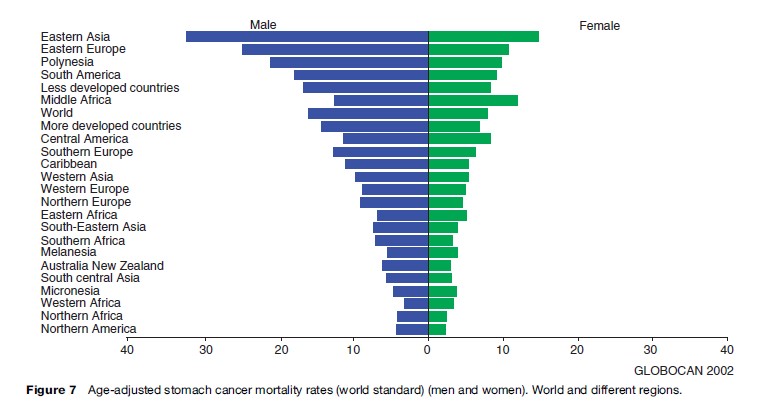
Stomach cancer was formerly the leading site of cancer deaths worldwide, but during the last several decades there has been a uniformly declining trend in stomach cancer mortality in most countries worldwide. This trend is probably due to a declining incidence of Helicobacter pylori infections in many populations along with better nutrition, including a need for less salt and salt-preserved food, alongside the higher relative consumption of fresh fruits and vegetables.
Liver Cancer
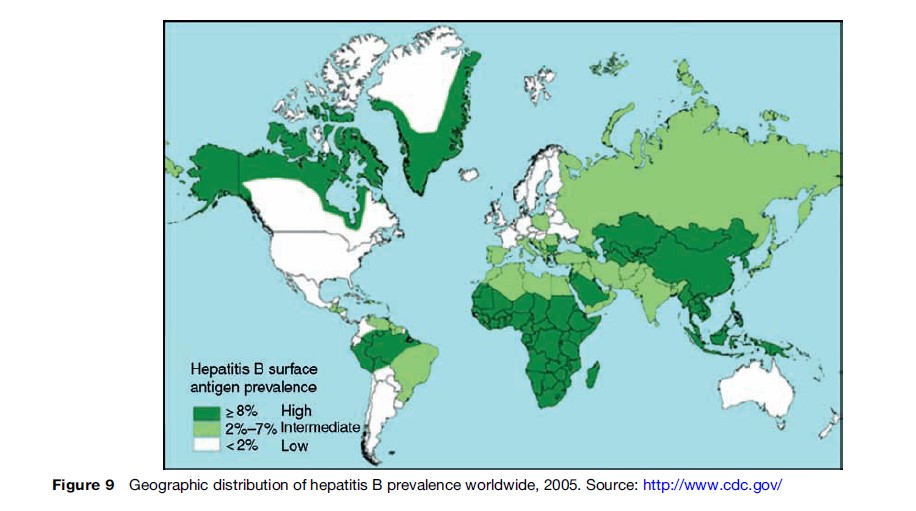
Liver cancer is a highly fatal neoplasm, with 5-year survival estimated at around 5% (Spangenberg et al., 2006). Nearly 600 000 people were estimated to have died of the disease in 2002, making it the third most common cause of death from cancer worldwide (GLOBOCAN, 2002). A large proportion of these deaths can be attributed to chronic infection with hepatitis B (Parkin, 2006), and the mortality largely follows the distribution of this infection agent (Figures 9, 10). Hepatitis C infection and consumption of foods contaminated by aflatoxin (WHO, 2006) are also important risk factors.
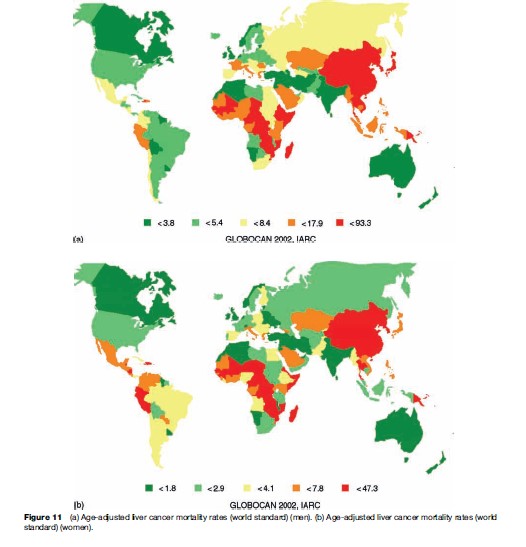
Liver cancer mortality is highest in Mongolia and Mozambique, with age-adjusted mortality rates of 93.3 and 78.4, respectively, per 100 000 for men, and 47.3 and 41.8, respectively, per 100 000 for women, whereas in South Central Asia, age-adjusted liver cancer mortality is 2.5 per 100 000 for men and 1.4 per 100 000 for women.
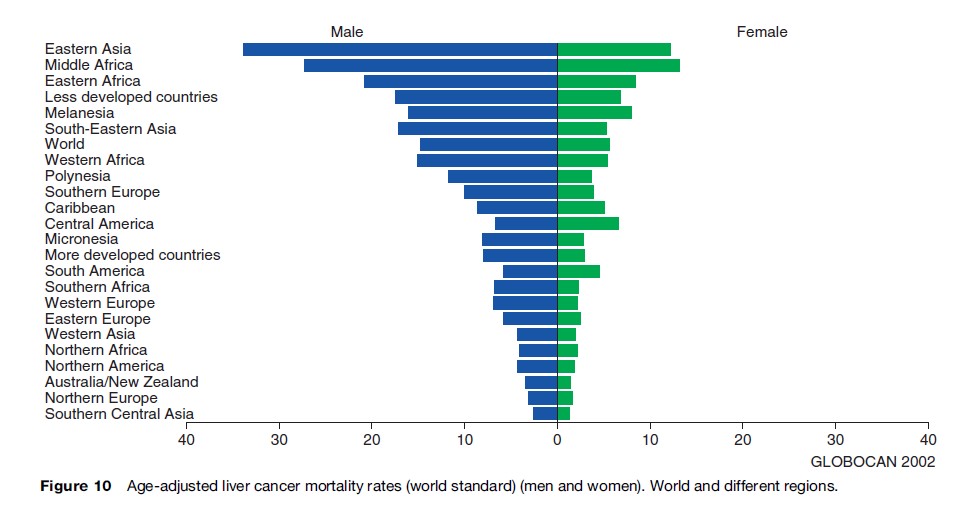
The age-adjusted liver cancer mortality is generally higher in developing than in developed countries. More than 80% of all liver cancer deaths occur in the less developed parts of the world.
Liver cancer mortality rates are considerably higher in men than women (Figure 11(a), (b)). In fact, the global age-adjusted liver cancer mortality rate among men is more than 2.5 times as high as for women: 14.9 and 5.7 per 100 000, respectively.
Consideration of the temporal variation of liver cancer mortality is particularly problematical for liver cancer. Mortality data may be unreliable because of the variable inclusion of metastatic liver cancers. There also have been changes in the ICD rubric in successive classifications, with the ICD code for liver cancer including gallbladder neoplasms in the seventh revision, only cancers of the liver and intrahepatic bile ducts specified as primary in the eighth revision, and including tumors that are unspecified (either primary or secondary) in the ninth revision.
Assessing Progress Against Cancer
Establishing whether a society is winning or losing the battle against cancer, and the relative merits and prioritization of primary, secondary, and tertiary prevention, have received much attention (in the United States in particular). Trends in cancer mortality rates have been at the forefront of the debate; the direction of trend has been considered by some as ‘the’ indicator of progress, pointing toward success or failure in controlling cancer, and informing the need to redress the balance of allocation of funding to preventative and treatment-orientated research.
Bailar and Smith’s paper in the New England Journal of Medicine in 1986 (Bailar and Smith, 1986), an assessment of the progress against cancer, based on trends in cancer mortality in the United States, 1950–82, was pessimistic in this respect, highlighting the failure of cancer control strategies to reduce the long-term mortality burden, and specifically, the need to shift research efforts away from treatment – given its perceived lack of impact on the mortality trend – to prevention. The conclusions were largely drawn from the fact that the overall age-adjusted mortality rates for all cancers combined had not changed appreciably over three decades. As the authors concluded, ‘‘by making deliberate choices among the measures, one can convey any impression from overwhelming success against cancer to disaster’’ (Doll et al., 1990: 501.)
Indeed, several authors were critical of some aspects of the study and a number of subsequent papers sought to readdress the issue. Sir Richard Doll (1999), in his assessment of progress in Europe, indicated that while mortality from all cancers was the correct outcome, the use of the all-age-adjusted rate was ‘‘profoundly wrong,’’ in that it outweighs the effect of recent progress with the ‘‘prevalence of carcinogenic agents in the distant past, which are irrelevant’’ (Doll et al., 1994). Any progress among young people, therefore, as would have been seen in the age-specific rates, would have been masked on combining age groups, and the notable successes (in high-resource settings) of therapeutic improvements for several relatively uncommon cancers, as discussed above, largely ignored.
Deliberations on progress against cancer, and the capability of different strategies to control the disease on the basis of past, present, and anticipated future mortality trends undoubtedly are set to continue in both scientific and political arenas well into the future. However, evaluating trends in many countries is clearly problematic given the paucity of data in many areas of the developing world as well as concerns as to the quality of the existing data.
Predicted Cancer Mortality In 2020
We end this research paper by predicting cancer mortality at the global level in 2020, demonstrating the profound impact that forecasted demographic changes will have on the predicted global number of cancer deaths. The planning of services is an integral component of cancer control programs, and predictions may inform us of the extent to which the determinants of the disease, and interventions, planned or unplanned, are likely to impact on the frequency of cancer in the years that follow. Investigation of the root determinants of the underlying trends is critically important in translating predictions to policy evaluation. Artifacts related to the data source and coding issues, the possible effect of interventions, and the etiological profile of the cancer type all need to be investigated.
Incorporating exposure data in making predictions may be considered among the best approaches; these are difficult to implement, however, given the present lack of understanding of the factors that drive most cancer trends, as well as a lack of availability of such data where determinants are established. As with incidence, however, time trends of cancer mortality rates cannot be accurately ascertained in many regions of the world, and it is not possible to speculate with any reasonable accuracy future burden. Thus even the use of extrapolation techniques involving recent cancer mortality trends is not readily achievable, and in making these predictions, we therefore make the crude assumption that cancer mortality rates will remain constant in the future.
The magnitude of the global cancer mortality burden will substantially increase in the next decades. In men, population aging and growth will mean a projected 55% increase in cancer deaths from the 3.8 million deaths estimated in 2002 to 5.9 million by 2020, even if overall rates remain the same. A similar increase in the cancer mortality burden is projected for women, with numbers of deaths expected to rise from 2.9 million in 2002 to 4.4 million in 2020, a 51% increase in the number of worldwide cancer deaths.
Bibliography:
- Bailar JC 3rd and Smith EM (1986) Progress against cancer? New England Journal of Medicine 314: 1226–1232.
- Boyle P (1989) Relative value of incidence and mortality data in cancer research. Recent Results in Cancer Research 114: 41–63.
- Clemmesen J and Nielsen A (1982) Morbidity versus mortality. Journal of National Cancer Institute 69: 418–428.
- Devesa SS, Pollack ES, and Young JLJ (1984) Assessing the validity of observed cancer incidence trends. American Journal of Epidemiology 119: 274–291.
- Doll R and Peto R (1981) The causes of cancer: Quantitative estimates of avoidable risks of cancer in the United States today. Journal of the National Cancer Institute 66: 1191–1308.
- Doll R (1990) Are we winning the fight against cancer? An epidemiological assessment. EACR—Mu¨ hlbock memorial lecture. European Journal of Cancer 26: 500–508.
- Ferlay J, Bray F, Pisani P, and Parkin DM (2004) Globocan 2002: Cancer Incidence, Mortality and Prevalence Worldwide. IARC CancerBase No. 5. version 2.0. Lyon, France: IARC Press.
- Grulich AE, Swerdlow AJ, dos Santos Silva I, and Beral V (1995) Is the apparent rise in cancer mortality in the elderly real? Analysis of changes in certification and coding of cause of death in England and Wales, 1970–1990. International Journal of Cancer 63: 164–168.
- Mackay J and Eriksen M (2002) The Tobaco Atlas. World Health Organization, http://whqlibdoc.who.int/publications/2002/9241562099.pdf
- Mathers CD, Fat DM, Inoue M, Rao C, and Lopez AD (2005) Counting the dead and what they died from: An assessment of the global status of cause of death data. Bulletin of the World Health Organization 83: 171–177.
- Parkin DM (2006) The global health burden of infection-associated cancers in the year 2002. International Journal of Cancer 118: 3030–3044.
- Percy C, Stanek E, and Gloecker L (1981) Accuracy of cancer death certificates and its effect on cancer mortality statistics. American Journal of Epidemiology 71: 242–250.
- Percy C and Dolman A (1978) Comparison of the coding of death certificates related to cancer in seven countries. Public Health Reports 93: 335–350.
- Peto R, Lopez AD, Boreham J, et al. (1996) Mortality from smoking worldwide. British Medical Bulletin 52: 12–21.
- Pisani P, Parkin DM, Bray F, and Ferlay J (1999) Estimates of the worldwide mortality from 25 cancers in 1990. International Journal of Cancer 83: 18–29.
- Spangenberg HC, Thimme R, and Blum HE (2006) Serum markers of hepatocellular carcinoma. Seminars in Liver Disease 26: 385–390.
- https://www-dep.iarc.fr/ – International Agency on Research on Cancer (IARC).
- https://www.who.int/ – World Health Organization website.




