This sample drugs research paper on drug abuse and alcohol dependence among inmates features: 6700 words (approx. 22 pages) and a bibliography with 39 sources. Browse other research paper examples for more inspiration. If you need a thorough research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Overview
The massive increase in the number of individuals within the United States (USA) criminal justice system (CJS) in the past 30 years can be largely attributed to the “War on Drugs” campaign with drug-related arrests increasing more than fivefold from 1970 to 2005. Many of the incarcerated individuals have a history of substance use disorders (SUDs), and have reported a use of drugs within the past month prior to arrest. According to the US Bureau of Justice Statistics (BJS), in 2002, the prevalence of offenders who used drugs more than once a week for at least 1 month prior to incarceration exceeded 60 % (Karberg and James 2005) and approximately 50 % of those incarcerated at the federal, state, and local levels met DSM-IV criteria for drug abuse or dependence.
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
The large burden of drug and alcohol dependence within the US CJS complicates treatment programs since drug and alcohol dependency is highly associated with comorbid psychiatric disorders and chronic infectious diseases. Effective pharmacotherapy for opioid and alcohol dependence exists in the community; however, access to treatment in the US CJS remains inadequate. Less than 10 % of local jail inmates received treatment (Karberg and James 2005).
The public health implications of SUDs treatment for offenders are numerous. Incarceration provides an opportunity to initiate or continue treatment for drug and alcohol dependency among inmates, whereupon both the individual and the community can be greatly benefited upon release. In addition to treatment, strong linkages should be established to ensure continuity of care. Failure to link services for offenders upon release to the community may increase recidivism, relapse to substance abuse, and increase transmission of sexually transmitted diseases (STDs) including human immunodeficiency virus (HIV), and viral hepatitis B and C to the uninfected.
This research paper will concentrate mostly on the epidemiology, treatment, and public health implications of opioid and alcohol dependence among inmates in the US CJS. While abuse of others drugs, such as cocaine and methamphetamine, is highly prevalent among offenders, much of the clinical and epidemiological literature on incarcerated persons has been limited to research involving dependency on alcohol and opioids. Effective medically assisted therapy (MAT) for cocaine, crack, and methamphetamine dependence is still lacking, and to date, only behavioral interventions have been implemented to treat dependence for these substances. Conversely, there are FDA-approved pharmacotherapies and behavioral interventions for opioidand alcohol-dependent persons within the community, and some of these have been successfully implemented in some incarcerated settings across the USA. This research paper will discuss the strengths and limitations of various evidence-based practices of SUDs treatment administered in incarcerated settings.
Definition Of Substance Dependency
The Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) classifies substance dependency as displaying three out of the seven of the listed criteria in Table 1 in a span of 12 months. Substance abuse, as defined by DSM-IV, is demonstration of at least one of the criteria in Table 1 in a span of 12 months.

Epidemiology And Socio-Demographic Profile Of Drug And Alcohol Abuse And Dependence In The Criminal Justice System
Prevalence of drug abuse among offenders in the US CJS remains high at the county, state, and federal level. In 2005 alone, there were 1.65 million drug arrests. Among all US county jails, nearly 60 % of inmates reported a history of drug dependency at some point in their life and about half of all US inmates met DSM-IV criteria for drug dependency (Mumola and Karberg 2006).
The majority of those incarcerated are men; however, the incarceration rate for women has been increasing since the early 1990s. From 1990 to 1998, the number of women behind bars jumped 71 %. This escalation of incarcerated women was mostly attributed to a surge in the arrest of female drug users. In 2002, about 61 % of incarcerated women satisfied DSM-IV criteria for drug dependency, versus 54 % of incarcerated men in local jails (Karberg and James 2005).
Minorities, especially blacks and Latinos, are disproportionately represented in the criminal justice system. While only making up about a quarter of the US population, more than 60 % of the US criminal justice population is black or Latino. Conversely, white inmates have been reported as having a relatively higher prevalence of substance (alcohol and/or drug) abuse or dependence (78 %) compared to blacks (64 %) and Hispanics (59 %) (Karberg and James 2005).
Opioid Abuse And Dependence
Abuse and dependence to opioids continues to plague the USA. There are approximately 900,000 opioid-dependent persons within the USA. In 2004, there were approximately 1.2 million state prisoners, of whom 23.4 % had ever used heroin or opiates in their lifetimes, and 8.2 % of convicted inmates reported using heroin or other opiates 1 month prior to arrest (Mumola and Karberg 2006). Approximately 11 % of all male inmates, and 20 % of all female inmates reported using opioids daily in the 6 months prior to arrest. Since the 2000s, nonmedical abuse of prescription opioids has led to an upsurge in number of arrests. A nationwide survey from years 2002 to 2004 reported that 30 % of arrestees had used prescription opioids for nonmedical purposes.
Stimulant Abuse And Dependence
The prevalence of abuse of stimulants (cocaine/ crack and methamphetamines) among inmates in state and federal prisons exceeds that of marijuana, opioids, hallucinogens, and other illicit depressants combined. In 2004, approximately one half of all federal and state prisoners with a history of drug use reported using cocaine and/or crack (Mumola and Karberg 2006). Additionally, methamphetamine (METH) abuse is a growing problem in the USA. From 1997 to 2004, use of METH was the only drug that increased across all measures collected in a nationwide survey (Mumola and Karberg 2006). There are stark differences in METH use by race/ ethnicity. At the federal level, the prevalence of METH abuse among white, Hispanic, and blacks is 29 %, 5 %, and 1 %, respectively (Mumola and Karberg 2006). METH use also differs by gender. Among state inmates, 17 % of women used METH in the month prior to arrest, as opposed to 10 % of men (Mumola and Karberg 2006). Furthermore, a nationwide survey of a sample of local police officers revealed that METH was overwhelmingly their drug of concern compared to cocaine, marijuana, and heroin (48 % vs. 22 %, 22 %, and 3 % respectively). Unlike treatment for opioids and alcohol, there is no effective FDA-approved pharmacotherapeutic treatment yet for cocaine and METH dependency. This is particularly problematic, given the increasing abuse of methamphetamines and high prevalence of cocaine abuse among inmates in state and federal prisons.
Alcohol Abuse And Dependence
Alcohol use disorders (AUDs) encompass a wide variety of conditions including abuse, hazardous drinking, binge drinking, and dependence. Approximately 4 % of the US population meets criteria for alcohol dependence (Grant et al. 2004), while the prevalence of alcohol dependence in state jails, however, is roughly tenfold higher than in the general population (Mumola and Karberg 2006). In 2002, nearly 50 % of all inmates met criteria for alcohol abuse or dependence (Mumola and Karberg 2006). The prevalence of alcohol abuse or dependence among female inmates was 39 %, while half of all men met the criteria for dependence or abuse (Mumola and Karberg 2006). Whites were relatively more likely to abuse alcohol or be dependent (58.6 %), compared to blacks (42.7 %) and Hispanic inmates (41.8 %).
Management And Treatment Of Opioid Dependence Within The Criminal Justice System
Despite the proven effectiveness of MAT to treat substance dependence in the community, long-term implementation is still rare across US incarcerated settings. The two most commonly administered pharmacotherapeutic options to manage and treat opioid dependence are opioid substitution therapy (OST) in the form of methadone and buprenorphine. The other options are antagonists at the mu-receptor in two formulations: once daily oral naltrexone (ReVia®), or the once monthly extended-release depot formulation of naltrexone (Vivitrol). See Table 2 for a summary of available MATs for the treatment of opioid dependence.
Methadone
Methadone is a full opioid mu-receptor agonist that produces similar effects to morphine and heroin. In terms of public health benefits, methadone has been associated with a reduction in risky injection practices (thereby reducing transmission of blood-borne viruses), improvement in overall health, and reduction in mortality. Methadone maintenance therapy (MMT) is also regarded as extremely cost-effective, with more than 50 % of the benefits affecting individuals who do not use drugs. In spite of the evidence demonstrating the advantages of MMT, it still remains difficult to obtain for the majority of heroin users. Access to methadone still remains a challenge for the majority of opioiddependent persons in the USA for several reasons including long waiting lists and because some state Medicaid programs do not cover the cost of MMT.
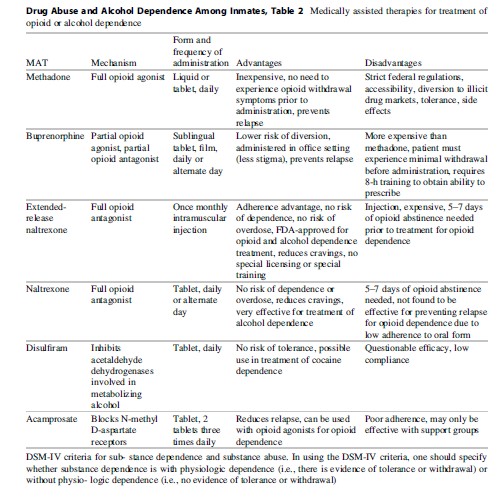
Authorities in the CJS still have strong reservations about offering methadone in prisons or jails. The predominating perception among criminal justice officials was that methadone itself “is replacing one addiction with another” (McMillan and Lapham 2005). Other reasons include concerns regarding cost, diversion, and overdose. Only a select few of incarcerated settings have set up the infrastructure necessary to implement a methadone maintenance treatment (MMT) program. Several research studies were conducted to investigate the effectiveness of the MMT program at Rikers Island (New York City) and found a decrease in risky injection practices 6 months post-release. Moreover, the implementation of a directly observed therapy approach of administration of methadone in the jail greatly alleviated correctional officials’ concern about possible diversion of methadone for illicit purposes (Tomasino et al. 2001). Furthermore, an RCT comparing (1) methadone after release, (2) methadone started in prison and transferred after release, and (3) counseling alone among opioid-dependent prisoners in Baltimore found that individuals who received only counseling were approximately seven times more likely to relapse to opioids and cocaine 12 months postrelease to the community, when compared to individuals who received both counseling and had initiated methadone in prison (Kinlock et al. 2009). Overall, both of these studies lend evidence that initiation of MMT is not only feasible within correctional institutions in the USA, but also that MMT has broad implications in reducing risky and illicit drug-using behavior when prisoners return to the community.
Buprenorphine
In 2002, the FDA approved buprenorphine (BPN), a partial mu-receptor agonist and antagonist for the treatment of opioid dependence. Providers need to take an 8-h course on BPN induction and maintenance practices to receive the necessary additional DEA license authorization to prescribe to patients. Since BPN can be administered in an office setting, there is less stigma associated with it compared to receiving daily doses of methadone at a federally sanctioned clinic. The dose response profile of BPN plateaus at a 32-mg dose (no additional effect for doses higher than 32 mg). Depending on the patient, BPN can be administered every 2–3 days, allowing for a more flexible maintenance regimen.
Due to its partial opioid agonist activity at the mu-receptor, BPN generally stimulates milder euphoric effects compared to methadone. BPN is most often administered sublingually, and the variant that includes naloxone (commercially known as Suboxone®) is thought to prevent misuse via injection of BPN. Recently, BPN has been introduced as a sublingual film that is more efficient in absorption than the tablet form. Furthermore, the chances of diversion are thought to be reduced due to a 10-digit bar code on the packaging that can be traced back to the patient should it be found in the illegal drug market and as a means to avoid accidental ingestion among minors.
Due to its relatively new arrival, research investigating the effectiveness of BPN in prison settings is still limited. Investigators conducted a RCT comparing BPN maintenance to MMT. The authors found that patients on BPN were more likely to attend community treatment centers compared to those randomized to receive methadone after release. Ninety-three (93) percent of the patients on BPN said that they intended to continue treatment after being released, while only 44 % of those on methadone expressed the same sentiment. While in jail, patients on BPN were significantly less likely to voluntarily drop out of receiving treatment than those on methadone. Despite these findings demonstrating more acceptability of BPN compared to methadone while the patients were incarcerated, there were no significant differences in terms of self-reported relapse to opioid use, rearrests, or reincarceration between the two groups. BPN was shown to reduce recidivism among inmates who initiated BPN while incarcerated compared to those who received no form of OST (Levasseur et al. 2002).
Implementation of a BPN maintenance program in a Baltimore prison highlighted some challenges distinct from MMT (Kinlock et al. 2010). The authors highlight diversion of BPN as the most significant challenge compared to methadone. Nurses had to wait 10 min per patient receiving a sublingual dose of BPN to ensure that the tablet had been completely dissolved. Methadone, on the other hand, is taken in liquid form, making diversion more difficult if the inmate is being directly observed. These findings are consistent with those from the Rikers Island study, with the authors reporting that approximately 10 % of the patients on BPN attempted to divert the drug, while only 1 % of the patients on methadone attempted diversion. Fears of diversion of BPN could be potentially mitigated if the sublingual film is used rather than the tablet, due to the former dissolving more rapidly than the latter. The FDA in 2012 ordered that tablet formulation of BPN would be replaced with the sublingual film in order to mitigate concerns for diversion and decrease deaths associated with inadvertent use of the sublingual tables by children.
Oral And Extended-Release Naltrexone
Buprenorphine and methadone are classified as types of opioid substitution therapy due to their affinity for the mu-receptor; however, naltrexone, a full opioid antagonist, completely blocks the mu-receptor. Naltrexone can be administered orally daily, or once a month by intramuscular injection (extended-release naltrexone). The oral form, ReVia®, approved in 1984 for the treatment of opioid dependence, has NOT been shown to be effective in relapse prevention or in recidivism due to compliance issues mainly. In October of 2010, the FDA approved the use of extended-release naltrexone (XR-NTX) for the treatment of opioid dependence after results from a double-blind multicenter RCT performed in Russia. In this study, 126 opioid-dependent individuals were randomized to receive the XR-NTX compared to 124 subjects who received placebo. The XR-NTX group was 1.58 times more likely to be abstinent from opioids at end of the 24-week study period compared to the control group. Of note, 40 % of the subjects who received XR-NTX had HIV disease, and approximately 90 % had Hepatitis C infection without any serious liver function abnormalities. The extended-release version of naltrexone does provide a possibility of less adherence concerns, given its once monthly dosing as well as possible fewer side effects compared to the oral form of naltrexone as well as methadone and BPN. Furthermore, because XR-NTX is an antagonist at the mu opioid receptor, there are no concerns for adverse events such as respiratory depression or overdose from the treatment as can be seen with methadone. Also due to its antagonist properties, there is not a concern for diversion as has been seen with buprenorphine. Therefore, XR-NTX may be a viable option for the CJS where such concerns predominate.
Before initiating naltrexone maintenance therapy, the patient must be free of opioids within the last 5–7 days. This is perhaps the most challenging aspect to naltrexone initiation, since many patients are fearful of experiencing any withdrawal symptoms and can often not resist the urge to continue using opioids in this period. While sudden abruption of not taking naltrexone does not produce withdrawal symptoms, the patient must be on oral naltrexone for at least 30 days to demonstrate efficacy in treatment outcomes with this form of naltrexone; however, it is unclear how long one should be maintained on XR-NTX at this time to demonstrate efficacy.
Thus far, only a few studies have examined the feasibility and effectiveness of oral or XRNTX maintenance therapy among incarcerated populations. In one double-blind RCT, investigators compared oral naltrexone to another opioid antagonist called cyclazocine among 40 inmates with prior history of opioid addiction (Brahen et al. 1977). At the end of the study, inmates who were on naltrexone reported fewer side effects after induction with a placebo and very low toxicity. In fact, three patients on cyclazocine dropped out of the study due to complications arising from the treatment itself. None of the patients randomized to naltrexone dropped out of the study for these reasons (Brahen et al. 1977). Indeed, a recent metaanalysis reported that the evidence claiming oral naltrexone as a superior treatment to other therapies for preventing relapse is conflicting (Minozzi et al. 2011).
There has been only one publication, thus far, on the effectiveness of XR-NTX among incarcerated populations. In this Norwegian study, 46 heroin-addicted inmates were randomized to either XR-NTX or methadone (Lobmaier et al. 2010). At 6 months post-release, the investigators found similar reductions in frequency of heroin use and criminal behavior between the two groups, suggesting that XR-NTX may be as equally effective as methadone at treatment of opioid relapse and recidivism. RCTs evaluating the effect of XR-NTX among opioid-dependent incarcerated populations are ongoing at the present time.
Behavioral Interventions For Offenders With Drug Dependence
Oftentimes pharmacotherapy is integrated with psychosocial support, either in the form of SUDs 12-step counseling, cognitive behavioral therapy, case management, or enrollment in therapeutic communities. Prison-based SUDs treatment programs have been shown to reduce recidivism and drug use. Specifically, cognitive behavioral therapy (CBT) interventions for offenders focusing on reducing criminal “thinking” can lead to criminal behavior. The objectives of CBT are to prevent relapse, to recognize situations that can often lead to drug use or criminal activities, to assist patients on how to effectively deal with these situations, to enhance social support networks, and to advance feelings of self-efficacy.
Contingency management (CM) is another type of behavioral intervention employed among drug abusing offenders that aims to promote abstinence through positive reinforcement. The theory supporting CM as an effective strategy is that rewarding positive behavior will eventually replace punishable behavior. Findings from two meta-analyses lend evidence to this theory (Griffith et al. 2000; Lussier et al. 2006). Modeled after community-based therapeutic communities, in-prison therapeutic communities (TCs) are a type of intervention that move drugusing offenders into separate facilities from the rest of the incarcerated population. This kind of residential treatment allows for intensive interventional and psychosocial support. The two TC modalities focus on the 12-step self-help model and the relapse prevention model. An evaluation of the Amity prison TC revealed statistically significant better outcomes in terms of reincarceration among inmates enrolled in TC versus inmates who dropped out of the program versus inmates randomized to a control group. Unlike OST, TCs have gained wide acceptance in the prison system. Since 1994, many states have adopted the TC model and received federal funding to expand residential substance abuse treatment (RSAT) programs in prisons and jails.
Management And Treatment Of Alcohol Dependence Within The Criminal Justice System
The prevalence of alcohol use disorders (AUDs) among incarcerated individuals is staggering, as previously discussed. Behavioral interventions have typically been viewed as the bastion for alcohol abuse treatment; however, these interventions have only been marginally effective in treating alcohol-related disorders (Springer et al. 2011). Specifically, behavioral treatment can include motivational enhancement therapy (MET), cognitive behavioral therapy, and self-help (e.g., 12-step) programs. MET, although less structured than CBT, has been shown to reduce cravings of alcohol with integrated pharmacotherapy using naltrexone (Monterosso et al. 2001). Currently, there are four FDA-approved pharmacotherapies available to treat alcohol dependence: naltrexone, extended-release naltrexone, acamprosate, and disulfiram. (These are summarized in Table 1).
Naltrexone
After being approved for opioid treatment by the FDA in 1984, oral naltrexone was approved 10 years later for the treatment of alcohol dependence after demonstrating a reduction in the pleasure produced from consumption of alcohol. Extended-release naltrexone (Vivitrol®) was FDA-approved for the treatment of alcohol dependence in 2006. Ethanol is believed to activate receptors in the opioid response system, which in turn activates numerous neurotransmitters, including dopamine. Since the efficacy of oral naltrexone is strongly contingent upon adherence, its usefulness as a treatment option for heavy drinkers has been questioned extensively. In many cases, compliance is the limiting factor in efficacy of oral naltrexone, while monthly injectable XR-NTX may overcome these challenges, due to fewer side effects and a better adherence profile (Mannelli et al. 2007). Although there has not yet been a head-to-head comparison of oral NTX and XR-NTX, evidence suggests that XR-NTX is highly efficacious for treatment of alcohol dependence, as well as compared to counseling or combined with counseling (O’Malley et al. 2007).
Disulfiram
Physiologically, disulfiram functions completely differently than naltrexone. Disulfiram blocks the oxidation of alcohol that results in increased levels of acetaldehyde. When a patient is administered disulfiram, yet continues to drink alcohol, the buildup of acetaldehyde can produce several negative effects such as nausea, vomiting, chest pain, and increased heart rate shortly after consumption. Similar to oral naltrexone, adherence to disulfiram is problematic unless the patients are highly motivated. Most noncontrolled trials among offenders demonstrated a beneficial effect of disulfiram, while one study did not (Bourne et al. 1966). One study among those on probation in Atlanta found 50 % of the subjects were abstinent from alcohol for over 3 months after daily observation of disulfiram administration (Bourne et al. 1966). Additionally, another study in New York Later studies, however, found that disulfiram was of no more benefit than placebo. One of these studies compared disulfiram to group therapy among alcoholic offenders in New Orleans and reported only a modest improvement in abstinence between the two groups.
Acamprosate
While both oral naltrexone and disulfiram have been available for decades, acamprosate has been available only since 2004. The biological mechanism of acamprosate’s activity is still not fully understood, yet it has been shown in numerous randomized clinical trials to maintain abstinence among alcohol dependents (who have already been detoxified from alcohol) modestly better than those who were administered placebo. In summary, as evidenced by findings from a large multicenter clinical trial known as the COMBINE study, oral naltrexone coupled with psychosocial support is still considered the most effective pharmacotherapeutic treatment for alcohol dependence (Anton et al. 2006; Garbutt et al. 2005).
Contraindications And Severe Adverse Events Of Naltrexone, Disulfiram, And Acamprosate
According to the manufacturer’s instructions, naltrexone should only be administered at the recommended doses. Liver injury has been documented when doses are excessive. Naltrexone should NOT be administered to patients with acute hepatitis or liver injury and is contraindicated in persons with childs-pugh class C cirrhosis. Naltrexone is also contraindicated in patients undergoing acute opioid withdrawal.
Disulfiram is contraindicated in patients receiving metronidazole, paraldehyde, alcohol, or alcohol-containing preparations (such as cough syrups). Patients with severe heart disease, coronary occlusion, and psychoses should avoid disulfiram. Disulfiram should never be administered to a patient currently under the influence of alcohol or without his/her knowledge. Several cases of hepatitis and liver failure have been documented in patients administered disulfiram.
Acamprosate is contraindicated in patients who have previously demonstrated hypersensitivity to acamprosate and in patients with severe kidney impairment. Similar to naltrexone, there has not been a rigorous evaluation on the safety of acamprosate in pregnant women.
The safety of naltrexone, disulfiram, and acamprosate has not been rigorously evaluated in pregnant women and is generally considered contraindicated in pregnancy. If the decision is made to use these drugs during pregnancy, careful consideration of the benefits and risks of administering these medications during pregnancy is necessary.
Medical And Psychiatric Comorbidities Associated With Dependency To Alcohol And Drug Abuse
Incarceration provides not only an opportunity to curb drug and alcohol use disorders, but also to address other underlying comorbidities. Infectious diseases, such as human immunodeficiency virus (HIV), tuberculosis (TB), viral hepatitis B (HBV) & C (HCV), and sexually transmitted infections (STIs) such as syphilis, gonorrhea, and chlamydia, are all strongly associated with drug and alcohol abuse. The burden of these diseases among individuals who have passed through the criminal justice system is disproportionately high, compared to those who have not. In 2006, it was estimated that one out of seven individuals living with HIV in the USA had been through the CJS or 16 % of the incarcerated population (Maruschak 2008; Spaulding et al. 2009). For HCV and TB, more than a third of all those infected in the USA were either incarcerated or just released from a correctional facility.
Human Immunodeficiency Virus (HIV)
The rate of HIV and AIDS among those incarcerated is approximately three times and four times higher respectively than the US general population (Maruschak 2008, 2009; Spaulding et al. 2009). The high rates of HIV among incarcerated persons are in part due to the overlap of increased frequency of alcohol and drug use among persons who are incarcerated and the high-risk injection-drug-using behaviors and unprotected sexual behaviors that occur under the influence of these substances. Concurrent opioid and alcohol use is significantly associated with higher morbidity and mortality, decreased adherence to highly active antiretroviral therapy (HAART), and increased transmission of HIV among HIV positive CJS populations. Among those with HIV disease who have comorbid alcohol or opioid dependence, adherence to HAART is low (Springer and Altice 2005). Thus, treatment of their chemical dependence should be prioritized among HIV-positive individuals in order to maintain adherence to HAART. In a small study among HIV-positive opioid-dependent incarcerated offenders in Connecticut, administration of both BPN and HAART resulted in low opioid positivity in urinalysis and high HIV viral load suppression at 3 months post-release (Springer et al. 2010; Springer et al. 2012). Currently, evaluation of administration of HIV treatment and MAT among incarcerated offenders before release is an active area of investigation.
Hepatitis C Virus (HCV)
With prevalence rates ranging from 16 % to 49 %, HCV is the most common blood-borne disease in the CJS. Injection-drug use is a strong risk factor for HCV acquisition. Most individuals with chronic HCV do not develop any symptoms; however, alcohol abuse can greatly accelerate end-stage liver disease (ESLD) and death (Springer et al. 2011). HCV treatment typically has been expensive and logistically difficult to implement in correctional settings due to its long duration. Newer and more effective treatments such as boceprevir and telaprevir that have shown more sustained virologic response when compared to standard treatment of pegylated interferon-ribavirin offer potential improved treatment opportunities for the incarcerated population as well. The CJS should consider evaluating the cost-effectiveness of HCV treatment, given the immense disease burden among inmates and potential risk to the community upon release.
HIV/HCV Coinfection
The prevalence of HIV/HCV coinfection is estimated to be between 50 % and 90 % among injection-drug users (IDUs). Estimates of prevalence of IDU among incarcerated adults range from 3 % to 28 %, and high-risk behaviors, such as sharing syringes, persist behind bars (Hammett 2006). Despite the potential for HIV and HCV acquisition in incarcerated settings, needle exchange programs are prohibited in American prisons and jails. Given the high burden of comorbid HIV and HCV disease in prisoners and the notion that ESLD is now the number one cause of death in HIV-positive persons, it behooves the CJS to consider treatment of HCV as well as treatment of drug and alcohol dependency to improve adherence to HCV treatment as a measure to decrease morbidity and mortality.
Tuberculosis (TB)
Transmission of TB is associated with unstable living conditions, poverty, and history of injection-drug use and is facilitated in crowded environments such as prisons and jails (Altice et al. 2010). The resurgence of TB in the last three decades can be largely attributed to the emergence of HIV (Altice et al. 2010). Susceptibility to TB is particularly high among those with high HIV viral loads due to suppression of the immune system from the virus (Friedland 2010). It has been estimated that in any given year, 40 % of all TB-positive individuals in the USA have circulated through the CJS (Hammett et al. 2002). At intake, all inmates are screened for TB. Given the high prevalence of drug and alcohol users who are TB positive in correctional settings, treatment of TB is imperative. First-line treatments for latent TB infection, such as rifampin and isoniazid, are highly effective (95 % cure rate) with successful adherence (Friedland 2010). Incarceration provides an opportunity where patients can be treated by directly observed therapy (DOT) after patients have detoxed from alcohol or drug use. Chronic alcohol use in conjunction with TB treatment can cause severe hepatotoxicity. Moreover, once released into the community without proper linkage to health care services, resumption of alcohol or drug can disrupt compliance to treatment regimens and promote drug-resistant strains of TB (Friedland 2010). Therefore, it is imperative that alcohol treatment be instituted in concert with effective TB treatment to prevent adherence interruptions and liver toxicity.
Sexually Transmitted Diseases (STDs)
Previous research has shown that drugs and alcohol are associated with risky sexual behaviors that increase the likelihood of transmission of HIV and other STDs. HIV and other STDs disproportionately affect women within the CJS. Women are more likely to be incarcerated for crimes related to drugs or sex work, two behaviors that are strong correlates of STD transmission. Additionally, incarcerated adult women have high rates of chlamydia (6.3 %), syphilis (7.5 %), while juvenile women in detention facilities have high rates of gonorrhea (5.7 %). Screening and treatment of STDs in incarcerated settings should be administered in addition to treatment for opioid and alcohol dependency. Such interventions could prevent jails and prison from becoming incubators of STDs, given the strong association between drug/alcohol dependency, risky sexual behavior, and incarceration.
Mental Illness
In addition to the large burden of infectious disease comorbidities among drug offenders, substantial proportions also suffer from severe mental illness. Schizophrenia, major depression, and bipolar disorder are all highly associated with substance abuse. In 1998, it was reported that nearly a quarter of federal inmates, and over a third of jail inmates with psychiatric disorders, had a history of alcohol dependence (Ditton 1999). Upon release, individuals with comorbid mental illnesses often fare poorly in both mental illness treatment and substance abuse treatment programs due to the lack of integrated care in addressing both these ailments in one health center. Thus, untreated comorbid psychiatric disorders place released drug offenders at high risk for criminal behavior, parole violation, and consequent reincarceration.
Linkage To The Community Post-Release
The majority of CJS populations will eventually be released to the community. One of the greatest challenges facing recently released offenders is relapse to drug or alcohol use. Nearly 90 % of individuals with opioid dependence not on drug treatment will relapse within 12 months after being released (Kinlock et al. 2002). Moreover, overdose is the number one cause of death among all released incarcerated persons in Washington state (Binswanger et al. 2007). Additionally, relapse is correlated with criminal behavior and reincarceration.
During this critical transition period, a robust framework must be in place to coordinate access to drug treatment, mental health services, HIV care, or other medical services once an inmate returns to the community. Case management services are seen as an effective linkage between the criminal justice system and the community. Various case management models exist; however, several share core principles such as client advocacy, monitoring, referral, and planning. Several case management interventions have been able to successfully link substance users to treatment upon release (Lincoln et al. 2006), but participation in case management services offered in correctional settings still remains a significant barrier to its effectiveness. While in some instances inmates may be mandated to utilize these services as conditions of their parole, those who volunteer to enroll in case management services often build stronger rapport with their case managers. Case managers are not viewed as representatives of the CJS; thus, clients are more willing to “open up” to them about illicit behavior. By establishing trust with their clients, case managers can advocate more efficiently on behalf of them and ensure greater retention with their services. Overall, it is not conclusive whether case management alone is sufficient to improve the health or curb drug use of released prisoners.
Future Directions
The “revolving door,” in reference to repeat offenders with problematic drug and alcohol abuse, continues to plague the US CJS and imposes a heavy burden on taxpayer dollars. Correctional care in the form of drug and alcohol treatment among offenders should be prioritized in order to disrupt this cycle. The very high influx of drug and alcohol dependents cycling through the CJS should be seen as an opportunity to initiate treatment and rehabilitation. In conjunction with behavioral interventions, effective pharmacotherapy has been shown to sustain abstinence and reduce cravings for opioids and alcohol for recently released inmates. Des
pite this, it cannot be emphasized enough that several major hurdles must be overcome in order to efficiently deliver correctional substance abuse care for incarcerated persons. First, improving education of correctional staff would likely increase acceptance of opioid and alcohol pharmacotherapies among correctional staff. The percentage of CJS settings offering medication-assisted therapy in the USA is dismally low, despite evidence indicating positive outcomes for offenders. Furthermore with the approval of Vivitrol® for treatment of opioid dependence as well as alcohol dependence, treatment options are expanded for offenders and the opioid antagonist may be more appealing to the CJS, given its once monthly adherence benefit and no overdose or diversion concerns.
Second, participation in psychosocial support programs in jails or prisons should be increased. Psychosocial groups, especially 12-step programs such as AA, have been very successful in reducing relapse to alcohol and drug use as has therapeutic communities and CBI.
Third, before release, correctional health care providers and case managers should work in concert to ensure that adherence to medically assisted therapy and medications for other comorbidities is not jeopardized, particularly for HIV disease. Relapse to drug and alcohol use is a significant risk factor for noncompliance or treatment failure, yet rigorous follow-up and treatment of substance use disorders can decrease relapse and recidivism rates. See Fig. 1.
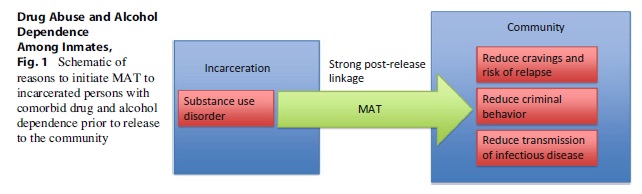
The evidence-based practices presented in this research paper are mostly limited to opioid and alcohol dependence. Polysubstance use is common among offenders, yet currently there is no federally approved medically assisted therapy for other highly abused addictive substances such as cocaine or methamphetamine. CBT has shown promising results in treating cocaine dependence; however, an effective pharmacologic treatment for long-term abstinence does not exist as of yet. Disulfiram, a pharmacotherapy for alcohol dependency, is being investigated as a possible treatment for reducing cocaine use. Additionally, clinical trials are currently being conducted to evaluate the efficacy of an anti-cocaine vaccine. In a trial with 115 individuals however, less than 40 % of subjects reached the requisite antibody level needed to block cocaine’s access to the brain. Treatment for methamphetamine dependency is of paramount importance now, as it is becoming a more emergent problem in many parts of the country. Potential immunotherapies for methamphetamine treatment have also been proposed; however, they still remain in the preclinical phase. Other pharmacotherapies show promise as well, such as XR-NTX and methylphenidate, but are still being evaluated.
Similar to management of most chronic diseases, treatment of substance use disorders among offenders requires a multidisciplinary approach inside and outside correctional settings. Moreover, a sizeable fraction of drug-using offenders suffer from numerous psychiatric and infectious comorbidities, which have broad societal and public health implications. Attention to inmates’ medical needs must be addressed in prisons and jails as a measure of safety, not just for the individual, but also from a public health standpoint.
Bibliography:
- Altice FL, Kamarulzaman A, Soriano VV, Schechter M, Friedland GH (2010) Treatment of medical, psychiatric, and substance-use comorbidities in people infected with HIV who use drugs. Lancet 376(9738):367–387. doi:10.1016/s0140-6736(10)60829-x
- Anton RF, O’Malley SS, Ciraulo DA, Cisler RA, Couper D, Donovan DM, Gastfriend DR, Hosking JD, Johnson BA, LoCastro JS, Longabaugh R, Mason BJ, Mattson ME, Miller WR, Pettinati HM, Randall CL, Swift R, Weiss RD, Williams LD, Zweben A (2006) Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA 295(17):2003–2017. doi:10.1001/jama.295.17.2003
- Binswanger IA, Stern MF, Deyo RA, Heagerty PJ, Cheadle A, Elmore JG, Koepsell TD (2007) Release from prison – a high risk of death for former inmates. N Engl J Med 356(2):157–165. doi:10.1056/ NEJMsa064115
- Bonczar TP, Glaze LE (2009) Publication probation and parole in the United States, 2008. US Department of Justice/Bureau of Justice Statistics, Washington, DC, NCJ 228230
- Bourne PG, Alford JA, Bowcock JZ (1966) Treatment of skid-row alcoholics with disulfiram. Q J Stud Alcohol 27(1):42–48
- Brahen LS, Capone T, Wiechert V, Desiderio D (1977) Naltrexone and cyclazocine. A controlled treatment study. Arch Gen Psychiatry 34(10):1181–1184
- Coviello DM, Cornish JW, Lynch KG, Alterman AI, O’Brien CP (2010) A randomized trial of oral naltrexone for treating opioid-dependent offenders. Am J Addict 19(5):422–432. doi:10.1111/j.1521-0391.2010.00070.x
- Ditton PM (1999) Mental health and treatment of inmates and probationers. US Department of Justice/Bureau of Justice Statistics, Washington, DC, NCJ 174463
- Friedland G (2010) Infectious disease comorbidities adversely affecting substance users with HIV: hepatitis C and tuberculosis. J Acquir Immune Defic Syndr 55(Suppl 1):S37–S42. doi:10.1097/ QAI.0b013e3181f9c0b6
- Garbutt JC, Kranzler HR, O’Malley SS, Gastfriend DR, Pettinati HM, Silverman BL, Loewy JW, Ehrich EW (2005) Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA 293(13):1617–1625. doi:10.1001/ jama.293.13.1617
- Grant BF, Dawson DA, Stinson FS, Chou SP, Dufour MC, Pickering RP (2004) The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991–1992 and 2001–2002. Drug Alcohol Depend 74(3):223–234. doi:10.1016/ j.drugalcdep.2004.02.004
- Griffith JD, Rowan-Szal GA, Roark RR, Simpson DD (2000) Contingency management in outpatient methadone treatment: a meta-analysis. Drug Alcohol Depend 58(1–2):55–66
- Hammett TM (2006) HIV/AIDS and other infectious diseases among correctional inmates: transmission, burden, and an appropriate response. Am J Public Health 96(6):974–978. doi:10.2105/ajph.2005.066993
- Hammett TM, Harmon MP, Rhodes W (2002) The burden of infectious disease among inmates of and releasees from US correctional facilities, 1997. Am J Public Health 92(11):1789–1794
- Karberg JC, James DJ (2005) Substance dependence, abuse, and treatment of jail inmates, 2002. US Department of Justice/Bureau of Justice Statistics, Washington, DC, NCJ 209588
- Kinlock TW, Battjes RJ, Schwartz RP (2002) A novel opioid maintenance program for prisoners: preliminary findings. J Subst Abuse Treat 22(3):141–147
- Kinlock TW, Gordon MS, Schwartz RP, Fitzgerald TT, O’Grady KE (2009) A randomized clinical trial of methadone maintenance for prisoners: results at 12 months postrelease. J Subst Abuse Treat 37(3):277–285. doi:10.1016/j.jsat.2009.03.002
- Kinlock TW, Gordon MS, Schwartz RP, Fitzgerald TT (2010) Developing and implementing a new prison-based buprenorphine treatment program. J Offender Rehabil 49(2):91–109. doi:10.1080/10509670903534951
- Levasseur L, Marzo JN, Ross N, Blatier C (2002) Frequency of re-incarcerations in the same detention center: role of substitution therapy. A preliminary retrospective analysis. Ann Med Interne (Paris) 153(3 Suppl):1S14–1S19
- Lincoln T, Kennedy S, Tuthill R, Roberts C, Conklin TJ, Hammett TM (2006) Facilitators and barriers to continuing healthcare after jail: a community-integrated program. J Ambul Care Manage 29(1):2–16
- Lobmaier PP, Kunoe N, Gossop M, Katevoll T, Waal H (2010) Naltrexone implants compared to methadone: outcomes six months after prison release. Eur Addict Res 16(3):139–145. doi:10.1159/000313336
- Lussier JP, Heil SH, Mongeon JA, Badger GJ, Higgins ST (2006) A meta-analysis of voucher-based reinforcement therapy for substance use disorders. Addiction 101(2):192–203. doi:10.1111/j.1360-0443.2006.01311.x
- Magura S, Lee JD, Hershberger J, Joseph H, Marsch L, Shropshire C, Rosenblum A (2009) Buprenorphine and methadone maintenance in jail and post-release: a randomized clinical trial. Drug Alcohol Depend 99(1–3):222–230. doi:10.1016/ j.drugalcdep.2008.08.006
- Mannelli P, Peindl K, Masand PS, Patkar AA (2007) Long-acting injectable naltrexone for the treatment of alcohol dependence. Expert Rev Neurother 7(10):1265–1277. doi:10.1586/14737175.7.10.1265
- Maruschak L (2008) HIV in Prisons, 2006 (NCJ 222179). US Department of Justice, Bureau of Justice Statistics, Washington, DC
- Maruschak L (2009) HIV in prisons, 2007–08. US Department of Justice/Bureau of Justice Statistics, Washington, DC
- McMillan GP, Lapham SC (2005) Staff perspectives on methadone maintenance therapy (MMT) in a large southwestern jail. Addict Res Theory 13(1):53–63. doi:10.1080/16066350512331328159
- Minozzi S, Amato L, Vecchi S, Davoli M, Kirchmayer U, Verster A (2011) Oral naltrexone maintenance treatment for opioid dependence. Cochrane Database Syst Rev (4), CD001333. doi:10.1002/14651858. CD001333.pub4. http://www.thecochranelibrary.com/ view/0/index.html
- Monterosso JR, Flannery BA, Pettinati HM, Oslin DW, Rukstalis M, O’Brien CP, Volpicelli JR (2001) Predicting treatment response to naltrexone: the influence of craving and family history. Am J Addict 10(3):258–268
- Mumola C (1999) Substance abuse and treatment of state and federal prisoners, 1997. US Department of Justice/ Bureau of Justice Statistics, Washington, DC, NCJ 172871
- Mumola C, Karberg JC (2006) Drug use and dependence, state and federal prisoners, 2004. US Department of Justice/Bureau of Justice Statistics, Washington, DC, NCJ 213530
- O’Malley SS, Garbutt JC, Gastfriend DR, Dong Q, Kranzler HR (2007) Efficacy of extended-release naltrexone in alcohol-dependent patients who are abstinent before treatment. J Clin Psychopharmacol 27(5):507–512. doi:10.1097/jcp.0b013e31814ce50d
- Rinaldo D (2008) Medicaid financing of medication-assisted treatment for opiate addiction. Accessed 28 Jan 2013
- Spaulding AC, Seals RM, Page MJ, Brzozowski AK, Rhodes W, Hammett TM (2009) HIV/AIDS among inmates of and releasees from US correctional facilities, 2006: declining share of epidemic but persistent public health opportunity. PLoS One 4(11):e7558. doi:10.1371/journal.pone.0007558
- Springer SA, Altice FL (2005) Managing HIV/AIDS in correctional settings. Curr HIV/AIDS Rep 2(4):165–170
- Springer SA, Chen S, Altice FL (2010) Improved HIV and substance abuse treatment outcomes for released HIV-infected prisoners: the impact of buprenorphine treatment. J Urban Health 87(4):592–602. doi:10.1007/s11524-010-9438-4
- Springer SA, Azar MM, Altice FL (2011) HIV, alcohol dependence, and the criminal justice system: a review and call for evidence-based treatment for released prisoners. Am J Drug Alcohol Abuse 37(1):12–21. doi:10.3109/00952990.2010.540280
- Springer SA, Qiu J, Saber-Tehrani AS, Altice FL (2012) Retention on buprenorphine is associated with high levels of maximal viral suppression among HIVinfected opioid dependent released prisoners. PLoS One 7(5):e38335. doi:10.1371/journal.pone.0038335
- Tomasino V, Swanson AJ, Nolan J, Shuman HI (2001) The Key Extended Entry Program (KEEP): a methadone treatment program for opiate-dependent inmates. Mt Sinai J Med 68(1):14–20




