View sample cancer research paper on breast cancer. Browse other research paper examples for more inspiration. If you need a thorough research paper written according to all the academic standards, you can always turn to our experienced writers for help. This is how your paper can get an A! Feel free to contact our writing service for professional assistance. We offer high-quality assignments for reasonable rates.
Breast cancer is one of the major public health problems. Every year some 1.2 million new cases of breast cancer are diagnosed and some 400 000 women die from it. Worldwide, some 50 million women are living after a previous breast cancer experience. Approximately 200 000 deaths from breast cancer occur in developed countries and 200 000 in developing countries. In Europe, there were an estimated 370 000 new cases and 130 000 deaths in 2004. Mortality rates rose from 1951 to about 1990 but have fallen since then in most European countries, noticeably in the UK (Figure 1(a)), but mortality rates in Central and Eastern European countries have been rising (Figure 1(b)). Although rates in Hong Kong and Japan have been lower than those in Europe, they have also been increasing (Figure 1(b)). Rates in North and South America have been similar to those in Western Europe (Figure 1(c)).
Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% OFF with 24START discount code
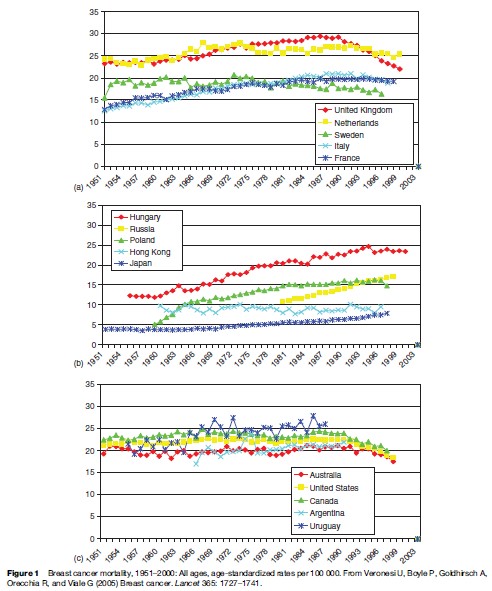
The decline in mortality rates in Western Europe, Australia, and the Americas may be due to widespread mammographic screening, good diagnosis, and increased numbers of women receiving the best treatment, including hormonal drugs (Ferlay et al., 2004).
Causes of Breast Cancer
Family History
In countries where breast cancer is common, the lifetime excess incidence of breast cancer is 5.5% for women with one first-degree relative who has had breast cancer and 13.3% for women with two. Eight out of nine women who develop the disease do not have an affected mother, sister, or daughter, however. Only 3–4% of women with breast carcinoma have a genetic mutation (BRCA1 or BRCA2), although these are the most strongly associated risk factors: Affected women have a 50–70% risk of developing breast carcinoma during their lifetime.
Pregnancy-Related And Hormone-Related Factors
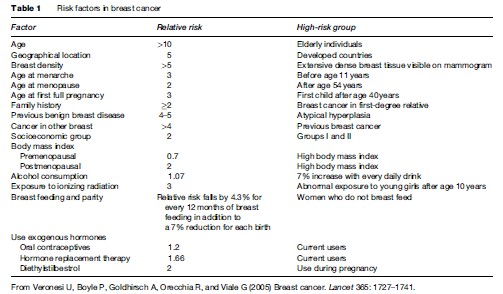
Women who have their first full-term birth at an early age have a lifetime reduction in risk. Increased parity is associated with a long-term risk reduction, even when the age at first birth is controlled for; the additional, long-lasting protective effect of a young age at subsequent full-term pregnancies is not as strong as that for the first full-term pregnancy; a nulliparous woman has roughly the same risk as a woman who has her first full-term birth aged about 30 years; the risk is transiently increased after a full-term pregnancy. Long duration of lactation confers a small, additional reduction in risk after the age at pregnancy and the number of full-term pregnancies is controlled for. Breast cancer risk factors are set out in Table 1.
The decision not to breastfeed or a very short lifetime duration of breastfeeding, typical of women in developed countries, contributes substantially to the high incidence of breast cancer in these areas. The risk is significantly reduced by breastfeeding, in addition to the reduction for every birth. Breastfeeding practices can be modified and promoted usefully as a strategy to prevent breast cancer.
There is no pronounced excess risk of diagnosis in women 10 or more years after the cessation of oral contraceptive use. The cancers diagnosed in women who have used combined oral contraceptives tend to be less advanced clinically than those diagnosed in women who have never used them. The risk of breast cancer is raised in women using hormone-replacement therapy and increases with duration of use. This effect declines after cessation of hormone-replacement therapy and largely disappears after about 5 years; the benefits and risks associated with this hormone treatment should be taken into account.
After adjustment for known risk factors, induced abortion is not associated with an increased risk of breast cancer. Pregnancies that end as a spontaneous or induced abortion have been shown not to increase a woman’s risk of developing breast cancer (Veronesi et al., 2005).
Anthropometric Indices And Physical Activity
With pooled data from seven prospective studies (337 819 women and 4385 incident breast cancer cases in total) and after adjustment for reproductive, dietary, and other risk factors, the pooled relative risk of breast cancer per height increment of 5 cm was 1.02 (95% CI 0.96–1.10) in premenopausal women and 1.07 (1.03–1.12) in postmenopausal women. The body mass index showed substantial inverse and positive associations with the disease in premenopausal and postmenopausal women, respectively. Height is an independent risk factor for breast cancer after menopause but not in premenopausal women.
In postmenopausal women not taking exogenous hormones, general obesity is an important predictor of breast cancer. In premenopausal women, weight and body mass index showed nonsignificant inverse associations with breast cancer.
Increased physical activity seems to be inversely related to the risk of breast cancer, although the findings are inconsistent. Physical activity and weight control can be recommended at present, although further research may highlight additional benefits.
Dietary Factors
A pooled analysis of eight prospective studies showed relative risks for an increment of 5% of energy intake were 1.09 for saturated fat, 0.93 for monounsaturated fat, and 1.05 for polyunsaturated fat, compared with equivalent energy intake from carbohydrates.
The Nurses’ Health Study II (Cho et al., 2003) showed that intake of animal fat, mainly from red meat, before menopause, was associated with an increased risk of breast cancer. To assess the risk of invasive breast cancer associated with total and beverage-specific alcohol consumption and to establish whether dietary and nondietary factors change such an association, data from six prospective studies were examined. Alcohol consumption correlated with breast cancer incidence in women. A reduction of consumption among women who drink alcohol regularly could reduce their risk of breast cancer. Cigarette smoking, frequently analyzed with alcohol consumption in etiological studies, does not seem to be related to risk.
Environmental Exposures
An increased risk of breast cancer in women exposed to ionizing radiation, particularly during puberty, has been widely accepted even with low-dose exposure. Environmental exposure to organochlorines has been examined as a potential risk factor for breast cancer. Based on current evidence, the association between risk and exposure to organochlorine pesticides and their residues seems to be small, if it exists at all. The combined evidence from five large U.S. studies that assessed the link between breast cancer risk and concentrations of 1,1-dichloro-2,2-bis (p-chlorophenyl) ethylene and polychlorinated biphenyls in blood plasma does not support such an association (Cuzick et al., 2003).
Possibilities Of Chemoprevention
The pharmacological prevention of cancer represents a comparatively novel field in clinical oncology, but it offers a very promising approach to reducing the burden of cancer and its incidence. In cardiology, it is common practice to treat subjects at higher risk for cardiovascular disease long before clinical evidence of the disease can be detected. This has made a definite contribution to lower mortality. A similar strategy could be adopted for cancer prevention in subjects ‘at higher risk’ (Hong and Sporn, 1997).
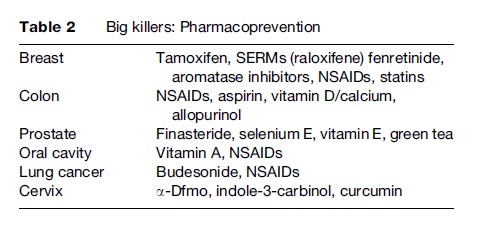
The peculiarity of carcinogenesis is that it is a multistep, multipath, and multifocal process, involving a series of genetic and epigenetic alterations that develop from genomic instability all the way to the final development of cancer. This is the key notion lying behind the rationale for intervention in the initial steps of the process, by employing natural or synthetic agents capable of delaying, arresting, or even reversing the pathogenesis of cancer. Since the process is generally very long (10–20 years, sometimes more), there is potentially a great deal of time to assess the true risk and intervene with nutrients and/or pharmacological agents that may interrupt the chain of molecular events long before the onset of clinical symptoms. This may prove of particular use where solid tumors are concerned, since they are often characterized by multifocality and metachronous growth. Recently, a number of compounds have been shown to be clinically effective in breast carcinoma, covering all three settings into which prevention may be typically divided. In primary prevention, the goal is to prevent the onset of the disease, selecting healthy cohorts who are at high risk because of their environment, lifestyle, or familial/ genetic factors. Secondary prevention (screening) is aimed at detecting and treating persons with a premalignant condition or an in situ malignancy, thus blocking its evolution to an invasive cancer. Tertiary prevention is a term that can be applied to the protection of individuals who have previously been treated for cancer from developing a second primary tumor (Figures 2 and 3; Table 2).
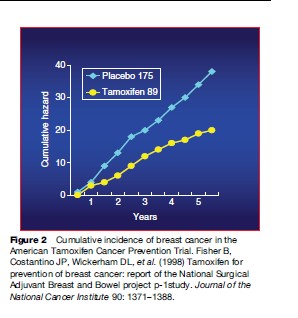
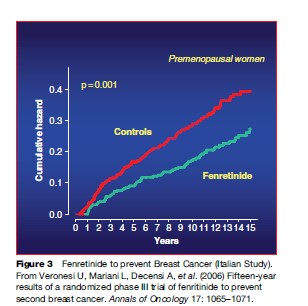
Pathogenesis of Breast Cancer
Progression From Healthy Tissue To Invasive Carcinoma
In contrast to the position for adenocarcinoma of the colon, no definitive model of progression from the common benign proliferative lesions of the breast to invasive malignancy has been identified. Cytological or architectural dysplastic changes can be identified in various nonmalignant breast diseases, such as florid and columnar duct hyperplasia, adenosis, and papilloma, but their true precancerous potential remains undefined.
Atypical duct hyperplasia has been regarded as the missing link between healthy duct hyperplasia and low-grade, ductal neoplasia in situ (DIN). Morphological features of atypical duct hyperplasia, which are intermediates of those in healthy and malignant tissues, and the substantially raised risk for subsequent carcinoma in affected women, have been claimed as sufficient proof of a precancerous nature. However, genetic changes in atypical duct hyperplasia are identical to changes in fully developed DIN, which raises questions about whether atypical duct hyperplasia is a distinct entity from low-grade intraductal neoplasia.
Novel approaches such as gene-expression profiling will increasingly be used to ascertain the occurrence of true preneoplastic lesions in the breast. Precise identification of these precursor lesions will be vital for planning interventions in women at high risk of breast cancer and to assess the effectiveness of prevention trials.
Ductal lavage is currently undergoing investigation. In this procedure, luminal cells from the ductal tree are obtained by cannulation of the lactiferous ducts and gentle massage of the breast. Harvested cells can then be examined not only for morphological changes but also for the expression of early markers of cell transformation that will eventually be identified. This procedure is being tested for validation as an additional research instrument to identify patients at high risk of developing breast carcinoma (O’Shaughnessy et al., 2002).
In view of the uncertainty of the occurrence of true preneoplastic lesions of the breast, the morphologically identifiable initial phases of neoplastic transformation remain in situ neoplasia, either ductal or lobular. However, this encompasses various lesions, ranging from low-grade to high-grade neoplasms, with remarkably different modes of presentation, histopathological features, genetic alterations, risk of relapse and of progression to invasive carcinoma.
To emphasize the non-life-threatening nature of in situ lesions and reduce any psychological effect caused by the use of carcinoma as a description, the term ductal intraepithelial neoplasia has been suggested to define these cell masses. This has been revised to encompass candidate preneoplastic lesions: Flat epithelial atypia and atypical duct hyperplasia (Tavassoli and Devilee, 2003). The same procedure has been done for noninvasive lobular neoplasms (atypical lobular hyperplasia and lobular carcinoma in situ), which have been classified into a three-tiered system of lobular intraepithelial neoplasia.
Invasion and metastasis are the hallmarks of fully developed breast carcinoma. Extensive histopathological examination of axillary sentinel lymph nodes by complete and serial sectioning at very close cutting intervals (e.g., at least 60 serial sections at 50-mm intervals, as used at the European Institute of Oncology, Milan, Italy) has greatly improved the detection rate of axillary lymphnode association (Veronesi et al., 2003). Detection is strongly associated with the definitive features of the primary breast cancer, such as tumor size and type, occurrence of peritumoral vascular invasion, and multifocality.
Randomized trials (Veronesi et al., 2003) have shown that the recorded number of patients with clinically overt axillary progression of breast cancer is much lower than expected, based on the false-negative rate of the sentinel lymph-node biopsy. This difference suggests that metastatic cells may not progress to clinical disease in all patients and that only some cells are able to sustain tumor progression, which is consistent with the hypothesis that the growth, progression, and clinical outcome of a cancer depend on the activation of tumorigenic stem/ progenitor cells.
This redefinition of the malignancy of breast cancer is recognized in the new pTNM classification, whereby minimum nodal invasion (1 mm or less) is classed as pT1mic (indicating microinvasive cancer) and isolated tumor cells or tumor-cell clusters (0.2 mm or less) in the regional lymph node are no longer regarded as metastatic, and qualify as pN0(iþ). These new classifications are designed to prevent overstaging of the disease and hence overtreatment for the patient.
Systemic adjuvant therapy is currently offered to patients according to selected clinicopathological features of the primary tumor, which include the status of estrogen and progesterone receptors and expression of human epidermal-growth-factor receptor 2 (HER2/neu); such treatment is undertaken independently of the axillary node status, with an equivalent survival benefit.
Quantification of tumor cells circulating in the blood of patients with breast cancer may also be a predictor of the duration of survival.
Gene-expression profiling of breast carcinoma has already shown that differential expression of specific genes is a more powerful prognostic indicator than traditional determinants such as tumor size and lymph-node status. These molecular assays now await clinical validation by prospective randomized trials before being introduced into routine clinical practice.
Breast Cancer Diagnosis and Staging
Organized screening, education programs, and improved consciousness of the female population have substantially changed the type of patients seen nowadays compared with those a few decades ago. The revolution in diagnostic imaging over the past 20 years has also profoundly modified diagnostic strategies in breast cancer.
Diagnostic Procedures
Procedures commonly used for the diagnosis of breast cancer include mammography, ultrasonography, MRI (magnetic resonance imaging), and PET (positron emission tomography). Physical examination of the breast remains important, however, because a substantial minority (11%) of breast cancers are not seen on mammography.
Mammography remains the most important diagnostic tool in women whose breast tissue is not dense. After menopause, mammography is generally the best method to discover tiny, nonpalpable lesions. By contrast, ultrasonography is the most effective procedure to diagnose small tumors in women with dense breast tissue and to differentiate solid lesions from cystic lesions. Although mammography can identify suspicious microcalcifications, it is not good at distinguishing between breast densities and has difficulty in identifying certain lobular invasive carcinomas, Paget’s disease of the nipple, inflammatory carcinoma, and particularly peripheral, small carcinomas.
MRI is mainly used as a problem-solving method after conventional diagnostic procedures. It is highly sensitive and is used mainly to screen high-risk, BRCA-positive patients. In dynamic, contrast-enhanced MRI, images are acquired before and after patients are given a substance to improve contrast when imaging the lesion. Malignant lesions are generally highly permeable, with rapid uptake and elimination of contrast, whereas benign lesions have slow-rising, persistent-enhancement kinetics. Although MRI has good diagnostic accuracy, the false-positive detection rate is still high and MRI findings should not be the sole indication for breast surgery.
PET is currently used to detect metastatic foci in any distant organ or to assess the status of axillary nodes in preoperative staging. However, PET may fail to identify low-grade lesions and tumors smaller than 5 mm.
The use of imaging techniques to detect unknown breast cancers in women who had no symptoms (i.e., screening) was inaugurated by the Health Insurance Plan of New York in the 1960s. In many randomized studies and population studies, mammography has been confirmed as the only screening test that can reduce breast cancer mortality if a large proportion of the population uses the procedure.
However, ultrasonography seems promising for women with dense breast tissue, such as those before menopause, and MRI has been valuable in the screening of women at high risk of breast cancer who are younger than 50 years.
Staging
The TNM (tumor-nodes-metastasis) system defines the extent of disease. It is the language used to compare different cases from various centers. With respect to the primary carcinoma (T), T1 can be divided into three subgroups (T1a, T1b, T1c), depending on the size of the primary lesion. However, with new subdivisions, most instances arise in one subcategory (e.g., T1c). In the era of computerized data analysis, classification is thought to be less necessary, whereas precise description of specific cases is regarded as essential and functional to the different needs of statisticians. Therefore, the T classification will probably be determined by a continuous metric description of the size (cm) of the carcinoma (e.g., T0.9, T2.4). The same system could apply to nodes (N) in which the numbers of involved and examined nodes will define the patient’s condition (e.g., N2/18 for two lymph nodes with tumor out of 18 sampled).
Finally, we believe that the TNM system should rely more on biological characteristics (e.g., hormonal receptors, proliferation rates) and biomolecular aspects (e.g., gene expression profile) of tumors. The present biometric, anatomical description will probably be replaced by molecular staging.
Breast Cancer Surgery
Once a diagnostic procedure indicates a tumor in the breast, cytological or histological confirmation is vital before further treatment is given. Cytology is effective in solid lesions, especially if sonographically guided. The histology of the lesion, which can be obtained by a core biopsy, is most useful for surgeons. This is the simplest method for palpable lesions that are easily reached, whereas a vacuum-assisted needle biopsy can obtain enough material for a good histological diagnosis in nonpalpable or deep lesions (Burbank et al., 1996). Excision biopsy a few days before definitive surgery is no longer done, because it creates a local anatomical distortion that makes conservative treatment difficult.
The sophisticated technique of sentinel lymph node biopsy provides knowledge about the condition of the axillary nodes without the need for dissection, when lymph nodes are not affected. Internal mammary nodes can also be easily reached during surgery, to complete the staging procedure. With respect to distant, occult metastases, PET will help identify occult foci of cancer cells anywhere in the body.
Breast conservation is the most popular treatment because most carcinomas have a restricted size and large primary tumors can be reduced in size by primary (neoadjuvant) chemotherapy before surgery. In most breast cancer centers, conservative surgery (lumpectomy) accounts for 75–85% of all operations. Total removal of the mammary gland (mastectomy) is required for multicentric invasive carcinoma, extensive intraduct carcinoma, inflammatory carcinoma, and large primary carcinomas that have not been sufficiently shrunk by neoadjuvant chemotherapy. Early recurrences or even a second ipsilateral carcinoma of restricted size can also be treated with conservative surgery.
Several options are available for reconstruction of the breast, from simple positioning of an expander to the use of musculocutaneous flaps (such as the thoracodorsal or abdominal flap, TRAM). One method becoming widely used is the skin-sparing mastectomy that conserves an extensive section of skin, as well as the more recent skin and nipple-sparing mastectomy, which preserves the nipple-areolar complex.
Identical 5-year survival has been recorded in women with axillary dissection and in women who underwent axillary dissection only if the sentinel lymph node was affected by tumor, although other clinical trials of the long-term effect on survival are ongoing. The histological diagnosis of the sentinel node should be immediately available. The traditional frozen-section procedure (which takes three or four sections of the node) often fails to detect micrometastases. As a consequence, surgeons should completely and definitively examine the sentinel node during surgery, and accurately section the node (up to 60–80 sections) to avoid missing even very small micrometastases. In about 85% of cases in which a micrometastatic sentinel node is found, other axillary nodes are not implicated. Therefore, many surgeons now consider the option of simply monitoring patients carefully with ultrasonography and PET.
In situ lesions are mainly treated with mammary resection. Since axillary metastases are rare, both lymph node dissection and biopsy are optional.
In situ neoplasia should not be incorporated in the TNM classification. Instead, it should be described with the new ductal-intraepithelial-neoplasia system proposed by Tavassoli (Tavassoli et al., 2003).
Radiotherapy in Breast Cancer
Radiotherapy In Breast Conservation
The current standard of care for patients with early-stage breast cancer consists of breast-conserving surgery, followed by 5–6 weeks’ postoperative radiotherapy. The need for radiotherapy in breast conservation is still debated. Some subgroups of patients may be expected to have a low risk of local recurrence, and radiotherapy could therefore be avoided. Attempts have been made to identify these populations, which might include individuals with small, low-grade tumors that are estrogenrecept-or-positive, or elderly patients resected with wide tumor-free margins, but no subgroup has been identified that would be adequately treated by breast-conserving surgery alone.
Radiotherapy is used on the whole breast. Some data support the effectiveness of an additional dose applied to the tumor bed (i.e., boost irradiation) to reduce local recurrence. The EORTC study results suggest that the patients deemed to receive the greatest absolute benefit from boost doses are those younger than 50 years and at higher risk of local recurrence (large tumor size or positive or small tumor-free margins in the surgical specimen).
Different radiation treatment schedules with rapid fractionation have been used for years in centers in the UK and Canada. Results from a randomized trial support delivery of a reduced total dose in a shortened schedule (42.5 Gy in 16 fractions for 22 days) in patients with lymph-node-negative breast cancer treated by lumpectomy. A short schedule (20 fractions) with concurrent use of the boost dose is currently used at the European Institute of Oncology Milan after quadrantectomy. In patients younger than 48 years who receive an intraoperative boost dose of 12 Gy, a rapid course of external radiotherapy is used (13 fractions of 2.85 Gy each).
Partial Breast Irradiation
The rationale for partial breast irradiation (restricted to the excision site and adjacent tissues) instead of the conventional approach is based on the finding that most recurrences arise near the primary tumor location. Partial breast irradiation can be delivered by different techniques, such as low or high-dose-rate brachytherapy (delivered interstitially or with an intracavitary balloon), conformal external-beam irradiation (including intensity-modulated radiotherapy), and intraoperative radiotherapy.
Intraoperative Radiotherapy
ELIOT (ELectron Intra Operative Therapy) refers to the application of a high dose of radiation during surgical intervention, after removal of the tumor.
ELIOT is currently used in early-stage breast cancer as the only treatment at the European Institute of Oncology, and a prospective randomized trial is ongoing. Two miniaturized mobile-linear accelerators producing a variable range of electron energies are available. Apart from low costs, ELIOT is advantageous because it potentially overcomes problems related to the accessibility of radiotherapy centers after surgery and has a beneficial impact on the patient’s quality of life. ELIOT does not irradiate the skin or the other breast, and irradiation to the lung and the heart is greatly reduced. ELIOT can also be used to give boost doses: One boost of 10–15Gy in an intraoperative session will extend surgery by just 10–20 min and reduces the time for external treatment by 2 weeks. The TARGIT (TARGeted Intraoperative radio-Therapy) trial is based on the use of a low-energy radiography source to compare one fraction of radiotherapy with a conventional postoperative approach.
Radiotherapy For Ductal Carcinoma In Situ
The role of radiotherapy in ductal carcinoma in situ (DCIS) managed with conservative treatment has been defined by results from three randomized trials. Addition of radiotherapy reduced the local recurrence rate by about 50%, with no effect on survival, and women with positive margins benefited the most. Despite these positive data, the best management of DCIS is still controversial. In fact, according to an analysis of a database of more than 25 000 patients treated between 1992 and 1999, almost half the women did not undergo postoperative radiotherapy after breast-conserving surgery.
Development In Radiation Techniques
Nowadays, the target volume can be tailored to individuals, which reduces the dose to the lung on the same side as the affected breast, and to the heart, the other breast, and surrounding soft tissue. Intensity-modulated beam arrangement ensures a more homogeneous dose delivery. The increasing use of optic or electronic devices (or both) to monitor organ motion and daily setup variations guarantees the accuracy and safety of the delivery system.
Radiotherapy In Locally Advanced Carcinoma
Breast-conserving surgery followed by radiotherapy can be offered to patients with locally advanced disease who respond to induction chemotherapy. A study of 340 patients given this combined treatment had a total locoregional recurrence rate of only 9% at 5-year follow-up. Breast reconstruction after mastectomy has become a standard procedure, and postmastectomy radiotherapy might represent an obstacle to good aesthetic results because of radiation-related fibrosis. The use of radiotherapy after mastectomy is still controversial.
Radiotherapy Of Metastases
With metastases in the skeleton, short courses of irradiation can palliate symptoms and prevent fractures. Radiotherapy and diphosphonates can improve the efficacy of the treatment. Brain metastases or carcinomatous meningitis can be treated successfully: Radiotherapy after complete surgical resection can substantially improve control. Patients with one brain metastasis who can be treated with more aggressive therapies, including surgery and high-precision radiotherapy, are especially challenging. Stereotactic radiotherapy is also used in other secondary tumor sites such as the liver, lung, and soft tissues.
Systemic Treatment of Breast Cancer
Treatment Of Locally Advanced Disease
The presence of estrogen and progesterone receptors in tumor cells shown by immunohistochemical staining is a good predictor of endocrine-responsiveness. Staining for either receptor indicates a response to endocrine therapies. Chemotherapy is also effective in endocrineresponsive disease, but the chances of more extensive cell killing are lower for these tumors than for tumors that are unresponsive.
The distinction between disease lacking expression of steroid-hormone receptors and disease showing some presence of these receptors is associated with gene-expression profiling and with the clinical course. Recognition of such a distinction will require a fundamental shift away from reporting whether the receptor status is positive or negative, which is current practice in many laboratories, to the quantitative reporting of receptor determinations.
Overexpression of the epithelial growth factor receptor HER2/neu on tumor-cell membranes is a strong predictor for response to trastuzumab (Tripathy et al., 2004). Overexpression of both steroid-hormone receptors and HER2/neu has been postulated as a condition for selective resistance to tamoxifen, but less so to aromatase inhibitors in postmenopausal women, and not to tamoxifen combined with suppression of ovarian endocrine function.
Adjuvant Treatments
Adjuvant systemic therapy is given to attempt eradication of micrometastatic disease, which may still be present in all patients with invasive breast cancer. It aims to reduce relapse and increase survival. Postoperative adjuvant therapies cannot be checked for efficacy except with respect to long-term outcomes in a randomized trial. In contrast, the efficacy of systemic treatment given either before surgery (i.e., primary treatments for operable or locally advanced breast cancer) or for metastases, should enable evaluation of the effect of treatment after short-term treatment.
Adjuvant systemic treatments are usually offered to reduce the risk of relapse. An expected 10-year survival below 90% would justify the use of adjuvant chemotherapy. The major concern about adjuvant cytotoxic treatments is that they are offered to a large proportion of patients who are either cured by local treatments or who might have their small risk of relapse reduced by endocrine drugs alone.
Selection of adjuvant treatments is based on the distinction between endocrine-unresponsive and endocrineresponsive breast cancer.
Patients with endocrine-responsive disease are offered adjuvant systemic therapy based on endocrine treatments. High risk of relapse (with metastatic lymph nodes in the operated axilla or vascular invasion) can justify some chemotherapy being given before endocrine treatment in adjuvant therapy.
Premenopausal women with endocrine-responsive disease are usually offered tamoxifen, with or without suppression of ovarian function. Use of cytotoxic drugs before endocrine therapy is recommended only if the risk of relapse is very high. However, the role of both ovarian function suppression and chemotherapy is still uncertain for many of these patients, and trials are in progress. Aromatase inhibitors, which need ovarian function suppression, represent an additional treatment choice.
Women with endocrine-responsive disease after menopause are usually offered endocrine therapy with tamoxifen and increasingly with aromatase inhibitors. Efficacy in reduction of recurrence and mortality well beyond 5 years’ treatment (carryover effect) is the reason for its standard use. When prescribed, chemotherapy should be given before the start of tamoxifen treatment.
New alternatives for tamoxifen are available to treat postmenopausal women with endocrine-responsive disease after surgery, after 2–3 years of tamoxifen to complete standard duration, or after 5 years to further reduce risk of relapse (especially for patients at high risk of relapse) (Coombes et al., 2004). These alternatives include nonsteroidal (anastrozole and letrozole) and steroidal (exemestane) aromatase inhibitors. The IBCSG (International Breast Cancer Study Group) trial 18–98 or BIG 1–98, which compares tamoxifen and letrozole alone or in the two possible sequences, recently provided data on greatly improved disease-free survival for postmenopausal patients with endocrine-responsive disease who received letrozole compared with those who received tamoxifen.
Neoadjuvant (Primary) Systemic Treatments of Breast Cancer
Systemic primary treatment is usually offered to patients with large primary tumors and aims to reduce tumor size for breast-conserving surgery. With such treatment, physicians can also induce regression of axillary node metastases and obtain knowledge on the responsiveness of the disease to treatment.
Endocrine therapies for patients with endocrineresponsive disease showed an improved outcome for aromatase inhibitors compared with that for tamoxifen.
Endocrine-unresponsive disease and high proliferation rates (e.g., Ki67 expressed in 20% of tumor cells) are important predictors of complete pathological response to six courses of primary chemotherapy. Disease-free survival is substantially longer for patients with endocrineresponsive disease than for patients who do not express steroid-hormone receptors, even though patients with endocrine-unresponsive disease are at least four times more likely to obtain a pathological complete remission after primary chemotherapy (Colleoni et al., 2004).
Anthracyclines and taxanes are usually used for patients with both operable and locally advanced disease (Nowak et al., 2004). Anthracycline-based primary chemotherapy has been reported to yield a large proportion of responses in small-sized tumors with a high proliferation index (Ki67) or grade, and with simultaneous overexpression of HER-2/neu and topoisomerase II, whereas mutation of p53 has been associated with a reduced response rate to chemotherapy. Chemotherapy regimens that do not contain anthracycline (that have vinorelbine, platinum, and fluorouracil) were also reported to be effective, especially for patients with endocrine-unresponsive disease, with or without inflammatory features.
Conclusions
Breast cancer is the most common type of tumor in women in most parts of the world. Although stabilized in Western countries, its incidence is increasing in other continents. Prevention of breast cancer is difficult because the causes are not well known. We know of many risk factors such as nulliparity, late age at first pregnancy, little or no breastfeeding, which, however, are linked to the historic development of human society. On the contrary, a great effort is needed to improve early detection of the tumor. Screening programs among the female population should therefore be implemented. The early discovery of a small breast carcinoma leads to a very high rate of curability and entails very mild types of treatment, with preservation of the body image. Treatments are improving, but a strict interdisciplinary approach is essential. It is conceivable that in all countries specialized centers or units for breast cancer management should be set up.
Bibliography:
- Burbank F, Parker SH, and Fogarty TJ (1996) Stereotactic biopsy: Improved tissue harvesting with the Mammotome. American Surgeon 62: 738–744.
- Cho E, Spiegelman D, Hunter DJ, et al. (2003) Premenopausal fat intake and risk of breast cancer. Journal of the National Cancer Institute 95(14): 1079–1085.
- Colleoni M, Viale G, Zahrieh D, et al. (2004) Chemotherapy is more effective in patients with breast cancer not expressing steroid hormone receptors: A study of preoperative treatment. Clinical Cancer Research 10: 6622–6628.
- Coombes RC, Hall E, Gibson LJ, et al. (2004) Intergroup Exemestane Study. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. New England Journal of Medicine 350: 1081–1092.
- Cuzick J, Powles T, Veronesi U, et al. (2003) Overview of main outcomes in breast cancer prevention trials. Lancet 361: 296–300.
- Ferlay J, Bray F, Pisani P, and Parkin DM (2004) Globocan 2002: Cancer Incidence, Mortality and Prevalence Worldwide. IARC CancerBase No. 5, version 2 0. Lyon, France: IARC Press.
- Fisher B, Costantino JP, Wickerham DL, et al. (1998) Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel project p-1study. Journal of the National Cancer Institute 90: 1371–1388.
- Hong WK and Sporn MB (1997) Recent advances in chemoprevention of cancer. Science 278: 1073–1077.
- Nowak AK, Wilcken NR, Stockler MR, Hamilton A, and Ghersi D (2004) Systematic review of taxane-containing versus non-taxane containing regimens for adjuvant and neoadjuvant treatment of early breast cancer. Lancet Oncology 5: 372–380.
- O’Shaughnessy JA, Ljung BM, Dooley WC, et al. (2002) Ductal lavage and the clinical management of women at high risk for breast carcinoma: A commentary. Cancer 94: 292–298.
- Tavassoli FA and Devilee P (eds.) (2003) World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Breast and Female Genital Tract. Lyon, France: IARC Press.
- Tripathy D, Slamon DJ, Cobleigh M, et al. (2004) Safety of treatment of metastatic breast cancer with trastuzumab beyond disease progression. Journal of Clinical Oncology 22: 1063–1070.
- Veronesi U, Paganelli G, Viale G, et al. (2003) A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. New England Journal of Medicine 349: 546–553.
- Veronesi U, Boyle P, Goldhirsch A, Orecchia R, and Viale G (2005) Breast cancer. Lancet 365: 1727–1741.
- Veronesi U, Mariani L, Decensi A, et al. (2006) Fifteen-year results of a randomized phase III trial of fenritinide to prevent second breast cancer. Annals of Oncology 17: 1065–1071.




